AGR2, an endoplasmic reticulum protein, is secreted into the gastrointestinal mucus
- PMID: 25111734
- PMCID: PMC4128659
- DOI: 10.1371/journal.pone.0104186
AGR2, an endoplasmic reticulum protein, is secreted into the gastrointestinal mucus
Abstract
The MUC2 mucin is the major constituent of the two mucus layers in colon. Mice lacking the disulfide isomerase-like protein Agr2 have been shown to be more susceptible to colon inflammation. The Agr2(-/-) mice have less filled goblet cells and were now shown to have a poorly developed inner colon mucus layer. We could not show AGR2 covalently bound to recombinant MUC2 N- and C-termini as have previously been suggested. We found relatively high concentrations of Agr2 in secreted mucus throughout the murine gastrointestinal tract, suggesting that Agr2 may play extracellular roles. In tissue culture (CHO-K1) cells, AGR2 is normally not secreted. Replacement of the single Cys in AGR2 with Ser (C81S) allowed secretion, suggesting that modification of this Cys might provide a mechanism for circumventing the KTEL endoplasmic reticulum retention signal. In conclusion, these results suggest that AGR2 has both intracellular and extracellular effects in the intestine.
Conflict of interest statement
Figures
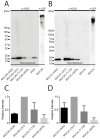
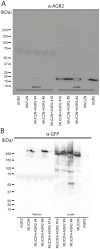



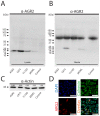
References
-
- Ermund A, Schutte A, Johansson MEV, Gustafsson JK, Hansson GC (2013) Studies of mucus in mouse stomach, small intestine, and colon. I. Gastrointestinal mucus layers have different properties depending on location as well as over the Peyer's patches. Am J Physiol Gastroint Liver Physiol 305: G341–G347. - PMC - PubMed
-
- Velcich A, Yang WC, Heyer J, Fragale A, Nicholas C, et al. (2002) Colorectal Cancer in Mice Genetically Deficient in the Mucin Muc2. Science 295: 1726–1729. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

