Low-level laser irradiation improves functional recovery and nerve regeneration in sciatic nerve crush rat injury model
- PMID: 25119457
- PMCID: PMC4131879
- DOI: 10.1371/journal.pone.0103348
Low-level laser irradiation improves functional recovery and nerve regeneration in sciatic nerve crush rat injury model
Abstract
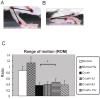
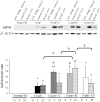
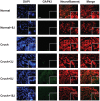
The development of noninvasive approaches to facilitate the regeneration of post-traumatic nerve injury is important for clinical rehabilitation. In this study, we investigated the effective dose of noninvasive 808-nm low-level laser therapy (LLLT) on sciatic nerve crush rat injury model. Thirty-six male Sprague Dawley rats were divided into 6 experimental groups: a normal group with or without 808-nm LLLT at 8 J/cm(2) and a sciatic nerve crush injury group with or without 808-nm LLLT at 3, 8 or 15 J/cm(2). Rats were given consecutive transcutaneous LLLT at the crush site and sacrificed 20 days after the crush injury. Functional assessments of nerve regeneration were analyzed using the sciatic functional index (SFI) and hindlimb range of motion (ROM). Nerve regeneration was investigated by measuring the myelin sheath thickness of the sciatic nerve using transmission electron microscopy (TEM) and by analyzing the expression of growth-associated protein 43 (GAP43) in sciatic nerve using western blot and immunofluorescence staining. We found that sciatic-injured rats that were irradiated with LLLT at both 3 and 8 J/cm(2) had significantly improved SFI but that a significant improvement of ROM was only found in rats with LLLT at 8 J/cm(2). Furthermore, the myelin sheath thickness and GAP43 expression levels were significantly enhanced in sciatic nerve-crushed rats receiving 808-nm LLLT at 3 and 8 J/cm(2). Taken together, these results suggest that 808-nm LLLT at a low energy density (3 J/cm(2) and 8 J/cm(2)) is capable of enhancing sciatic nerve regeneration following a crush injury.
Conflict of interest statement
Figures



References
-
- Navarro X, Vivo M, Valero-Cabre A (2007) Neural plasticity after peripheral nerve injury and regeneration. Prog Neurobiol 82: 163–201. - PubMed
-
- Allodi I, Udina E, Navarro X (2012) Specificity of peripheral nerve regeneration: interactions at the axon level. Prog Neurobiol 98: 16–37. - PubMed
-
- Navarro X, Vivó M, Valero-Cabré A (2007) Neural plasticity after peripheral nerve injury and regeneration. Progress in Neurobiology 82: 163–201. - PubMed
-
- Colen KL Choi M, Chiu DT (2009) Nerve grafts and conduits. Plast Reconstr Surg 124: e386–394. - PubMed
-
- Gu X, Ding F, Yang Y, Liu J (2011) Construction of tissue engineered nerve grafts and their application in peripheral nerve regeneration. Prog Neurobiol 93: 204–230. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

