Detection of AGEs as markers for carbohydrate metabolism and protein denaturation
- PMID: 25120273
- PMCID: PMC4078063
- DOI: 10.3164/jcbn.13-112
Detection of AGEs as markers for carbohydrate metabolism and protein denaturation
Abstract
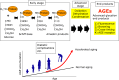
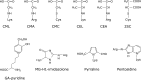
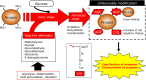
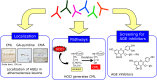
Approximately 100 years have passed since the Maillard reaction was first reported in the field of food chemistry as a condensation reaction between reducing sugars and amino acids. This reaction is thought to progress slowly primarily from glucose with proteins in vivo. An early-stage product, called the "Amadori product", is converted into advanced glycation end products. Those accumulate in the body in accordance with age, with such accumulation being enhanced by lifestyle-related diseases that result in the denaturation of proteins. Recent studies have demonstrated that intermediate carbonyls are generated by several pathways, and rapidly generate many glycation products. However, accurate quantification of glycation products in vivo is difficult due to instability and differences in physicochemical properties. In this connection, little is known about the relationship between the structure of glycation products and pathology. Furthermore, the interaction between proteins modified by glycation and receptors for advanced glycation end products is also known to induce the production of several inflammatory cytokines. Therefore, those inhibitors have been developed over the world to prevent lifestyle-related diseases. In this review, we describe the process of protein denaturation induced by glycation and discuss the possibility of using the process as a marker of age-related diseases.
Keywords: AGEs; aging; diabetic complications; glycation; post-translational modification.
Figures
Similar articles
-
In vitro kinetic studies of formation of antigenic advanced glycation end products (AGEs). Novel inhibition of post-Amadori glycation pathways.J Biol Chem. 1997 Feb 28;272(9):5430-7. doi: 10.1074/jbc.272.9.5430. J Biol Chem. 1997. PMID: 9038143
-
Kinetics of nonenzymatic glycation of ribonuclease A leading to advanced glycation end products. Paradoxical inhibition by ribose leads to facile isolation of protein intermediate for rapid post-Amadori studies.Biochemistry. 1996 Apr 16;35(15):4645-54. doi: 10.1021/bi9525942. Biochemistry. 1996. PMID: 8664253
-
Modification of proteins in vitro by physiological levels of glucose: pyridoxamine inhibits conversion of Amadori intermediate to advanced glycation end-products through binding of redox metal ions.J Biol Chem. 2003 Nov 21;278(47):46616-24. doi: 10.1074/jbc.M307155200. Epub 2003 Sep 15. J Biol Chem. 2003. PMID: 12975371
-
Pathologic role of dietary advanced glycation end products in cardiometabolic disorders, and therapeutic intervention.Nutrition. 2016 Feb;32(2):157-65. doi: 10.1016/j.nut.2015.08.001. Epub 2015 Sep 1. Nutrition. 2016. PMID: 26602289 Review.
-
[Therapeutic intervention in diseases with advanced glycation end products in their pathogenesis].Pol Merkur Lekarski. 2009 Aug;27(158):152-6. Pol Merkur Lekarski. 2009. PMID: 19856885 Review. Polish.
Cited by
-
Rapid formation of N ε-(carboxymethyl)lysine (CML) from ribose depends on glyoxal production by oxidation.RSC Chem Biol. 2024 Sep 18;5(11):1140-6. doi: 10.1039/d4cb00183d. Online ahead of print. RSC Chem Biol. 2024. PMID: 39323732 Free PMC article.
-
Generation and characterization of monoclonal antibody against Advanced Glycation End Products in chronic kidney disease.Biochem Biophys Rep. 2016 Mar 23;6:142-148. doi: 10.1016/j.bbrep.2016.03.011. eCollection 2016 Jul. Biochem Biophys Rep. 2016. PMID: 28955871 Free PMC article.
-
Causative or associative: A critical review of the role of advanced glycation end-products in bone fragility.Bone. 2022 Oct;163:116485. doi: 10.1016/j.bone.2022.116485. Epub 2022 Jul 4. Bone. 2022. PMID: 35798196 Free PMC article. Review.
-
Changes in S-(2-succinyl)cysteine and advanced glycation end-products levels in mouse tissues associated with aging.Amino Acids. 2022 Apr;54(4):653-661. doi: 10.1007/s00726-022-03130-y. Epub 2022 Feb 15. Amino Acids. 2022. PMID: 35166937
-
Advanced glycation and glycoxidation end products in bone.Bone. 2023 Nov;176:116880. doi: 10.1016/j.bone.2023.116880. Epub 2023 Aug 12. Bone. 2023. PMID: 37579812 Free PMC article. Review.
References
-
- Nakayama H, Taneda S, Mitsuhashi T, et al. Characterization of antibodies to advanced glycosylation end products on protein. J Immunol Methods. 1991;140:119–125. - PubMed
-
- Horiuchi S, Araki N, Morino Y. Immunochemical approach to characterize advanced glycation end products of the Maillard reaction. Evidence for the presence of a common structure. J Biol Chem. 1991;266:7329–7332. - PubMed
-
- Koito W, Araki T, Horiuchi S, Nagai R. Conventional antibody against Nepsilon-(carboxymethyl)lysine (CML) shows cross-reaction to Nepsilon-(carboxyethyl)lysine (CEL): immunochemical quantification of CML with a specific antibody. J Biochem. 2004;136:831–837. - PubMed
-
- Nagai R, Ikeda K, Higashi T, et al. Hydroxyl radical mediates Nε-(carboxymethyl)lysine formation from Amadori product. Biochem Biophys Res Commun. 1997;234:167–172. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

