Maternal control of the Drosophila dorsal-ventral body axis
- PMID: 25124754
- PMCID: PMC4724419
- DOI: 10.1002/wdev.138
Maternal control of the Drosophila dorsal-ventral body axis
Abstract
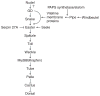
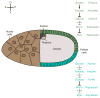
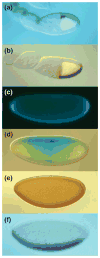
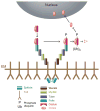
The pathway that generates the dorsal-ventral (DV) axis of the Drosophila embryo has been the subject of intense investigation over the previous three decades. The initial asymmetric signal originates during oogenesis by the movement of the oocyte nucleus to an anterior corner of the oocyte, which establishes DV polarity within the follicle through signaling between Gurken, the Drosophila Transforming Growth Factor (TGF)-α homologue secreted from the oocyte, and the Drosophila Epidermal Growth Factor Receptor (EGFR) that is expressed by the follicular epithelium cells that envelop the oocyte. Follicle cells that are not exposed to Gurken follow a ventral fate and express Pipe, a sulfotransferase that enzymatically modifies components of the inner vitelline membrane layer of the eggshell, thereby transferring DV spatial information from the follicle to the egg. These ventrally sulfated eggshell proteins comprise a localized cue that directs the ventrally restricted formation of the active Spätzle ligand within the perivitelline space between the eggshell and the embryonic membrane. Spätzle activates Toll, a transmembrane receptor in the embryonic membrane. Transmission of the Toll signal into the embryo leads to the formation of a ventral-to-dorsal gradient of the transcription factor Dorsal within the nuclei of the syncytial blastoderm stage embryo. Dorsal controls the spatially specific expression of a large constellation of zygotic target genes, the Dorsal gene regulatory network, along the embryonic DV circumference. This article reviews classic studies and integrates them with the details of more recent work that has advanced our understanding of the complex pathway that establishes Drosophila embryo DV polarity. For further resources related to this article, please visit the WIREs website.
Conflict of interest: The authors have declared no conflicts of interest for this article.
© 2014 Wiley Periodicals, Inc.
Conflict of interest statement
Conflict of interest: The authors have declared no conflicts of interest for this article.
Figures





References
-
- Roth S, Jordan P, Karess R. Binuclear Drosophila oocytes: consequences and implications for dorsal-ventral patterning in oogenesis and embryogenesis. Development. 1999;126:927–934. - PubMed
-
- Nüsslein-Volhard C. Maternal effect mutations that alter the spatial coordinates of the embryo of Drosophila melanogaster. In: Subtelny S, Konigsberg IR, editors. Determinants of Spatial Organization: the Thirty-Seventh Symposium of the Society for Developmental Biology. New York: Academic Press; 1979. pp. 185–211.
FURTHER READING
-
- Flybase; St Pierre SE, Ponting L, Stefancsik R, McQuilton P the FlyBase Consortium. Flybase 102 - advanced approaches to interrogating Flybase. Nucleic Acids Res. 2014;42(D1):D780–D788. doi: 10.1093/nar/gkt1092. Available at: http://www.flybase.org. - DOI - PMC - PubMed
-
- The interactive fly. [Accessed May 19, 2014];Brody TB. 2013 Available at: http://www.sdbonline.org/sites/fly/aimain/1aahome.htm.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

