The challenge and promise of anti-epileptic therapy development in animal models
- PMID: 25127174
- PMCID: PMC5003536
- DOI: 10.1016/S1474-4422(14)70076-6
The challenge and promise of anti-epileptic therapy development in animal models
Erratum in
- Lancet Neurol. 2015 Jul;14(7):677
Abstract
Translation of successful target and compound validation studies into clinically effective therapies is a major challenge, with potential for costly clinical trial failures. This situation holds true for the epilepsies-complex diseases with different causes and symptoms. Although the availability of predictive animal models has led to the development of effective antiseizure therapies that are routinely used in clinical practice, showing that translation can be successful, several important unmet therapeutic needs still exist. Available treatments do not fully control seizures in a third of patients with epilepsy, and produce substantial side-effects. No treatment can prevent the development of epilepsy in at-risk patients or cure patients with epilepsy. And no specific treatment for epilepsy-associated comorbidities exists. To meet these demands, a redesign of translational approaches is urgently needed.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of interests JE has a patent pending for a magnetonanoparticle for functional MRI; JE has received no fees to date. (WO2009/123734A1 and WO2009/123735A1). ASG has received royalties from Morgan & Claypool Publishers and John Libbey Eurotext, and consultancy honorarium from Viropharma. FEJ has received investigator-initiated grant support from Eisai Pharma and Lundbeck Pharma. SLM has received consultancy honorarium from Lundbeck and UCB Pharma. TO’B has received research funding from Sanofi-Aventis, UCB, SciGen, GlaxoSmithKline, Novartis, and Janssen-Cilag, and speakers’ honoraria from Sanofi-Aventis, UCB, SciGen, GlaxoSmithKline, and Janssen-Cilag. JAF is the president of The Epilepsy Study Consortium, a non-profit organisation; New York University (NYU) receives a fixed amount from the Epilepsy Study Consortium towards her salary. The money is for work by JAF on behalf of The Epilepsy Study Consortium, for consulting and clinical trial-related activities. She receives no personal income for these activities. Within the past year, The Epilepsy Study Consortium received payments for research services from: Acorda, Eisai Medical Research, GlaxoSmithKline, Impax, Johnson & Johnson, Mapp Pharmaceuticals, Marinus, Novartis, Lundbeck, Pfizer, Sepracor, Sunovion, SK Life Science, Supernus Pharmaceuticals, UCB/Schwarz Pharma, Upsher Smith, Vertex. JAF is an investigator at NYU on studies for Eisai Medical Research, LCGH, Impax, Mapp Pharmaceuticals, Novartis, UCB/Schwarz Pharma, Upsher Smith, and Vertex. MS, ARB-K, SLM, AP, and KSW declare no competing interests.
Figures
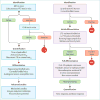

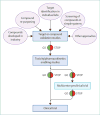
Comment in
-
Epilepsy: lost in translation.Lancet Neurol. 2014 Sep;13(9):862-3. doi: 10.1016/S1474-4422(14)70125-5. Epub 2014 Aug 10. Lancet Neurol. 2014. PMID: 25127172 No abstract available.
References
-
- Löscher W, Schmidt D. Modern antiepileptic drug development has failed to deliver: ways out of the current dilemma. Epilepsia. 2011;52:657–78. - PubMed
-
- Galanopoulou AS, Buckmaster PS, Staley KJ, et al. the American Epilepsy Society Basic Science Committee And The International League Against Epilepsy Working Group On Recommendations For Preclinical Epilepsy Drug Discovery Identification of new epilepsy treatments: issues in preclinical methodology. Epilepsia. 2012;53:571–82. - PMC - PubMed
-
- Steward O, Popovich PG, Dietrich WD, Kleitman N. Replication and reproducibility in spinal cord injury research. Exp Neurol. 2012;233:597–605. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 NS038595/NS/NINDS NIH HHS/United States
- R01 NS080565/NS/NINDS NIH HHS/United States
- U01 NS042372/NS/NINDS NIH HHS/United States
- R37 NS065434/NS/NINDS NIH HHS/United States
- R56 NS031718/NS/NINDS NIH HHS/United States
- U01 NS045911/NS/NINDS NIH HHS/United States
- R01 NS020253/NS/NINDS NIH HHS/United States
- R01 NS031718/NS/NINDS NIH HHS/United States
- R37 NS033310/NS/NINDS NIH HHS/United States
- P01 NS002808/NS/NINDS NIH HHS/United States
- R21 NS083057/NS/NINDS NIH HHS/United States
- P20 NS080181/NS/NINDS NIH HHS/United States
- R01 NS078331/NS/NINDS NIH HHS/United States
- R01 NS065434/NS/NINDS NIH HHS/United States
- R01 NS043209/NS/NINDS NIH HHS/United States
- R21 NS080268/NS/NINDS NIH HHS/United States
- R37 NS043209/NS/NINDS NIH HHS/United States
- U01 NS038455/NS/NINDS NIH HHS/United States
- R01 NS033310/NS/NINDS NIH HHS/United States
- R56 NS020253/NS/NINDS NIH HHS/United States
- R21 NS078333/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

