Biomechanical strain as a trigger for pore formation in Schlemm's canal endothelial cells
- PMID: 25128579
- PMCID: PMC4175173
- DOI: 10.1016/j.exer.2014.08.003
Biomechanical strain as a trigger for pore formation in Schlemm's canal endothelial cells
Abstract
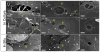
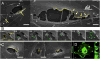
The bulk of aqueous humor passing through the conventional outflow pathway must cross the inner wall endothelium of Schlemm's canal (SC), likely through micron-sized transendothelial pores. SC pore density is reduced in glaucoma, possibly contributing to obstructed aqueous humor outflow and elevated intraocular pressure (IOP). Little is known about the mechanisms of pore formation; however, pores are often observed near dome-like cellular outpouchings known as giant vacuoles (GVs) where significant biomechanical strain acts on SC cells. We hypothesize that biomechanical strain triggers pore formation in SC cells. To test this hypothesis, primary human SC cells were isolated from three non-glaucomatous donors (aged 34, 44 and 68), and seeded on collagen-coated elastic membranes held within a membrane stretching device. Membranes were then exposed to 0%, 10% or 20% equibiaxial strain, and the cells were aldehyde-fixed 5 min after the onset of strain. Each membrane contained 3-4 separate monolayers of SC cells as replicates (N = 34 total monolayers), and pores were assessed by scanning electron microscopy in 12 randomly selected regions (∼65,000 μm(2) per monolayer). Pores were identified and counted by four independent masked observers. Pore density increased with strain in all three cell lines (p < 0.010), increasing from 87 ± 36 pores/mm(2) at 0% strain to 342 ± 71 at 10% strain; two of the three cell lines showed no additional increase in pore density beyond 10% strain. Transcellular "I-pores" and paracellular "B-pores" both increased with strain (p < 0.038), however B-pores represented the majority (76%) of pores. Pore diameter, in contrast, appeared unaffected by strain (p = 0.25), having a mean diameter of 0.40 μm for I-pores (N = 79 pores) and 0.67 μm for B-pores (N = 350 pores). Pore formation appears to be a mechanosensitive process that is triggered by biomechanical strain, suggesting that SC cells have the ability to modulate local pore density and filtration characteristics of the inner wall endothelium based on local biomechanical cues. The molecular mechanisms of pore formation and how they become altered in glaucoma may be studied in vitro using stretched SC cells.
Keywords: Schlemm's canal endothelium; biomechanics; endothelium; pore; strain; stretch.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures




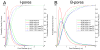
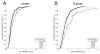
References
-
- Albelda SM, Sampson PM, Haselton FR, McNiff JM, Mueller SN, Williams SK, Fishman AP, Levine EM. Permeability characteristics of cultured endothelial cell monolayers. Journal of Applied Physiology. 1988;64:308–322. - PubMed
-
- Allingham RR, de Kater AW, Ethier CR, Anderson PJ, Hertzmark E, Epstein DL. The relationship between pore density and outflow facility in human eyes. Investigative Ophthalmology & Visual Science. 1992;33:1661–1669. - PubMed
-
- Battista SA, Lu Z, Hofmann SR, Freddo TF, Overby DR, Gong H. Reduction of the Available Area for Aqueous Humor Outflow and Increase in Meshwork Herniations into Collector Channels Following Acute IOP Elevation in Bovine Eyes. Investigative Ophthalmology & Visual Science. 2008;49:5346–5352. - PMC - PubMed
-
- Camras LJ, Stamer WD, Epstein D, Gonzalez P, Yuan F. Differential Effects of Trabecular Meshwork Stiffness on Outflow Facility in Normal Human and Porcine Eyes. Investigative Ophthalmology & Visual Science. 2012;53:5242–5250. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

