Non-natural acetogenin analogues as potent Trypanosoma brucei inhibitors
- PMID: 25145275
- PMCID: PMC4298241
- DOI: 10.1002/cmdc.201402272
Non-natural acetogenin analogues as potent Trypanosoma brucei inhibitors
Abstract
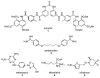
Neglected tropical diseases remain a serious global health concern. Here, a series of novel bis-tetrahydropyran 1,4-triazole analogues based on the framework of chamuvarinin, a polyketide natural product isolated from the annonaceae plant species are detailed. The analogues synthesized display low micromolar trypanocidal activities towards both bloodstream and insect forms of Trypanosoma brucei, the causative agent of African sleeping sickness, also known as Human African Trypanosomiasis (HAT). A divergent synthetic strategy was adopted for the synthesis of the key tetrahydropyran intermediates to enable rapid access to diastereochemical variation either side of the 1,4-triazole core. The resulting diastereomeric analogues displayed varying degrees of trypanocidal activity and selectivity in structure-activity relationship studies. Together, the biological potency and calculated lipophilicity values indicate that while there is room for improvement, these derivatives may represent a promising novel class of anti-HAT agents.
Keywords: Human African Trypanosomiasis (HAT); Trypanosoma brucei; acetogenin; natural product analogues; neglected diseases; stereochemistry.
© 2014 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures
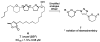
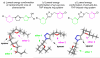





Similar articles
-
Development of Simplified Heterocyclic Acetogenin Analogues as Potent and Selective Trypanosoma brucei Inhibitors.ChemMedChem. 2016 Jul 19;11(14):1503-6. doi: 10.1002/cmdc.201600210. Epub 2016 Jun 10. ChemMedChem. 2016. PMID: 27283448 Free PMC article.
-
Simplifying nature: Towards the design of broad spectrum kinetoplastid inhibitors, inspired by acetogenins.Bioorg Med Chem. 2017 Nov 15;25(22):6126-6136. doi: 10.1016/j.bmc.2017.01.021. Epub 2017 Jan 28. Bioorg Med Chem. 2017. PMID: 28185724
-
Total synthesis, stereochemical assignment, and biological activity of chamuvarinin and structural analogues.Chemistry. 2013 Jun 17;19(25):8309-20. doi: 10.1002/chem.201204527. Epub 2013 Apr 29. Chemistry. 2013. PMID: 23630031
-
3-(Oxazolo[4,5-b]pyridin-2-yl)anilides as a novel class of potent inhibitors for the kinetoplastid Trypanosoma brucei, the causative agent for human African trypanosomiasis.Eur J Med Chem. 2013 Aug;66:450-65. doi: 10.1016/j.ejmech.2013.05.007. Epub 2013 May 16. Eur J Med Chem. 2013. PMID: 23831695
-
Novel 3-nitro-1H-1,2,4-triazole-based piperazines and 2-amino-1,3-benzothiazoles as antichagasic agents.Bioorg Med Chem. 2013 Nov 1;21(21):6600-7. doi: 10.1016/j.bmc.2013.08.022. Epub 2013 Aug 20. Bioorg Med Chem. 2013. PMID: 24012457 Free PMC article.
Cited by
-
Structure-Based Design, Synthesis and Biological Evaluation of Bis-Tetrahydropyran Furan Acetogenin Mimics Targeting the Trypanosomatid F1 Component of ATP Synthase.European J Org Chem. 2019 Sep 1;2019(31-32):5434-5440. doi: 10.1002/ejoc.201900541. Epub 2019 May 29. European J Org Chem. 2019. PMID: 31598093 Free PMC article.
-
Development of Simplified Heterocyclic Acetogenin Analogues as Potent and Selective Trypanosoma brucei Inhibitors.ChemMedChem. 2016 Jul 19;11(14):1503-6. doi: 10.1002/cmdc.201600210. Epub 2016 Jun 10. ChemMedChem. 2016. PMID: 27283448 Free PMC article.
-
A recent update on new synthetic chiral compounds with antileishmanial activity.Chirality. 2022 Oct;34(10):1279-1297. doi: 10.1002/chir.23494. Epub 2022 Aug 10. Chirality. 2022. PMID: 35947400 Free PMC article. Review.
-
Photo-affinity labelling and biochemical analyses identify the target of trypanocidal simplified natural product analogues.PLoS Negl Trop Dis. 2017 Sep 5;11(9):e0005886. doi: 10.1371/journal.pntd.0005886. eCollection 2017 Sep. PLoS Negl Trop Dis. 2017. PMID: 28873407 Free PMC article.
-
Uncovering the Mechanism of Action of Antiprotozoal Agents: A Survey on Photoaffinity Labeling Strategy.Pharmaceuticals (Basel). 2024 Dec 28;18(1):28. doi: 10.3390/ph18010028. Pharmaceuticals (Basel). 2024. PMID: 39861091 Free PMC article. Review.
References
-
- Trypanosomiasis, African. World Health Organization; Geneva: [Accessed 19 June, 2014]. 2012. http://www.who.int/topics/trypanosomiasis_african/en/
-
- Priotto G, Kasparian S, Mutombo W, Ngouama D, Ghorashian S, Arnold U, Ghabri S, Baudin E, Buard V, Kazadi-Kyanza S, Ilunga M, Mutangala W, Pohlig G, Schmid C, Karunakara U, Torreele E, Kande V. Lancet. 2009;374:56–64. - PubMed
-
- Alali FQ, Liu X-X, McLaughlin JL. J. Nat. Prod. 1999;62:504–540. - PubMed
- Zafra-Polo M-C, Figadère B, Gallardo T, Tormo JR, Cortes D. Phytochemistry. 1998;48:1087–1117.
- Zafra-Polo MC, González MC, Estornell E, Sahpaz S, Cortes D. Phytochemistry. 1996;42:253–271. - PubMed
- Zeng L, Ye Q, Oberlies NH, Shi G, Gu Z-M, He K, McLaughlin JL. Nat. Prod. Rep. 1996;13:275–306. - PubMed
- Rupprecht JK, Hui YH, McLaughlin JL. J. Nat. Prod. 1990;53:237–278. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

