Platelet concentration in platelet-rich plasma affects tenocyte behavior in vitro
- PMID: 25147809
- PMCID: PMC4132404
- DOI: 10.1155/2014/630870
Platelet concentration in platelet-rich plasma affects tenocyte behavior in vitro
Abstract
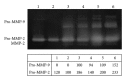
Since tendon injuries and tendinopathy are a growing problem, sometimes requiring surgery, new strategies that improve conservative therapies are needed. Platelet-rich plasma (PRP) seems to be a good candidate by virtue of its high content of growth factors, most of which are involved in tendon healing. This study aimed to evaluate if different concentrations of platelets in PRP have different effects on the biological features of normal human tenocytes that are usually required during tendon healing. The different platelet concentrations tested (up to 5 × 10(6) plt/µL) stimulated differently tenocytes behavior; intermediate concentrations (0.5 × 10(6), 1 × 10(6) plt/µL) strongly induced all tested processes (proliferation, migration, collagen, and MMPs production) if compared to untreated cells; on the contrary, the highest concentration had inhibitory effects on proliferation and strongly reduced migration abilities and overall collagen production but, at the same time, induced increasing MMP production, which could be counterproductive because excessive proteolysis could impair tendon mechanical stability. Thus, these in vitro data strongly suggest the need for a compromise between extremely high and low platelet concentrations to obtain an optimal global effect when inducing in vivo tendon healing.
Figures






References
-
- Pietrzak WS, Eppley BL. Platelet rich plasma: biology and new technology. Journal of Craniofacial Surgery. 2005;16(6):1043–1054. - PubMed
-
- Sommeling CE, Heyneman A, Hoeksema H, Verbelen J, Stillaert FB, Monstrey S. The use of platelet-rich plasma in plastic surgery: a systematic review. Journal of Plastic, Reconstructive & Aesthetic Surgery. 2013;66(3):301–311. - PubMed
-
- de Mos M, van der Windt AE, Jahr H, et al. Can platelet-rich plasma enhance tendon repair? A cell culture study. The American Journal of Sports Medicine. 2008;36(6):1171–1178. - PubMed
-
- Eppley BL, Woodell JE, Higgins J. Platelet quantification and growth factor analysis from platelet-rich plasma: implications for wound healing. Plastic and Reconstructive Surgery. 2004;114(6):1502–1508. - PubMed
-
- Giusti I, Rughetti A, D'Ascenzo S, et al. The effects of platelet gel-released supernatant on human fibroblasts. Wound Repair and Regeneration. 2013;21(2):300–308. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

