Evidence against translational repression by the carboxyltransferase component of Escherichia coli acetyl coenzyme A carboxylase
- PMID: 25157077
- PMCID: PMC4248805
- DOI: 10.1128/JB.02091-14
Evidence against translational repression by the carboxyltransferase component of Escherichia coli acetyl coenzyme A carboxylase
Abstract
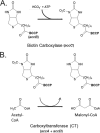
In Escherichia coli, synthesis of the malonyl coenzyme A (malonyl-CoA) required for membrane lipid synthesis is catalyzed by acetyl-CoA carboxylase, a large complex composed of four subunits. The subunit proteins are needed in a defined stoichiometry, and it remains unclear how such production is achieved since the proteins are encoded at three different loci. Meades and coworkers (G. Meades, Jr., B. K. Benson, A. Grove, and G. L. Waldrop, Nucleic Acids Res. 38:1217-1227, 2010, doi:http://dx.doi.org/10.1093/nar/gkp1079) reported that coordinated production of the AccA and AccD subunits is due to a translational repression mechanism exerted by the proteins themselves. The AccA and AccD subunits form the carboxyltransferase (CT) heterotetramer that catalyzes the second partial reaction of acetyl-CoA carboxylase. Meades et al. reported that CT tetramers bind the central portions of the accA and accD mRNAs and block their translation in vitro. However, long mRNA molecules (500 to 600 bases) were required for CT binding, but such long mRNA molecules devoid of ribosomes seemed unlikely to exist in vivo. This, plus problematical aspects of the data reported by Meades and coworkers, led us to perform in vivo experiments to test CT tetramer-mediated translational repression of the accA and accD mRNAs. We report that increased levels of CT tetramer have no detectable effect on translation of the CT subunit mRNAs.
Copyright © 2014, American Society for Microbiology. All Rights Reserved.
Figures

Similar articles
-
The Classical, Yet Controversial, First Enzyme of Lipid Synthesis: Escherichia coli Acetyl-CoA Carboxylase.Microbiol Mol Biol Rev. 2021 Aug 18;85(3):e0003221. doi: 10.1128/MMBR.00032-21. Epub 2021 Jun 16. Microbiol Mol Biol Rev. 2021. PMID: 34132100 Free PMC article. Review.
-
A tale of two functions: enzymatic activity and translational repression by carboxyltransferase.Nucleic Acids Res. 2010 Mar;38(4):1217-27. doi: 10.1093/nar/gkp1079. Epub 2009 Dec 3. Nucleic Acids Res. 2010. PMID: 19965770 Free PMC article.
-
Growth rate regulation of Escherichia coli acetyl coenzyme A carboxylase, which catalyzes the first committed step of lipid biosynthesis.J Bacteriol. 1993 Jan;175(2):332-40. doi: 10.1128/jb.175.2.332-340.1993. J Bacteriol. 1993. PMID: 7678242 Free PMC article.
-
[A novel gene (Aa-accA ) encoding acetyl-CoA carboxyltransferase alpha-subunit of Alkalimonas amylolytica N10 enhances salt and alkali tolerance of Escherichia coli and tobacco BY-2 cells].Wei Sheng Wu Xue Bao. 2013 Aug 4;53(8):809-16. Wei Sheng Wu Xue Bao. 2013. PMID: 24341272 Chinese.
-
Isoforms of acetyl-CoA carboxylase: structures, regulatory properties and metabolic functions.Biochem Soc Trans. 1997 Nov;25(4):1232-8. doi: 10.1042/bst0251232. Biochem Soc Trans. 1997. PMID: 9449982 Review. No abstract available.
Cited by
-
A series of medium and high copy number arabinose-inducible Escherichia coli expression vectors compatible with pBR322 and pACYC184.Plasmid. 2015 Sep;81:21-6. doi: 10.1016/j.plasmid.2015.03.001. Epub 2015 May 27. Plasmid. 2015. PMID: 26021570 Free PMC article.
-
Interaction of the Nitrogen Regulatory Protein GlnB (PII) with Biotin Carboxyl Carrier Protein (BCCP) Controls Acetyl-CoA Levels in the Cyanobacterium Synechocystis sp. PCC 6803.Front Microbiol. 2016 Oct 26;7:1700. doi: 10.3389/fmicb.2016.01700. eCollection 2016. Front Microbiol. 2016. PMID: 27833596 Free PMC article.
-
Phospholipid synthesis inside phospholipid membrane vesicles.Commun Biol. 2022 Sep 27;5(1):1016. doi: 10.1038/s42003-022-03999-1. Commun Biol. 2022. PMID: 36167778 Free PMC article.
-
The Classical, Yet Controversial, First Enzyme of Lipid Synthesis: Escherichia coli Acetyl-CoA Carboxylase.Microbiol Mol Biol Rev. 2021 Aug 18;85(3):e0003221. doi: 10.1128/MMBR.00032-21. Epub 2021 Jun 16. Microbiol Mol Biol Rev. 2021. PMID: 34132100 Free PMC article. Review.
-
Reassessment of the Genetic Regulation of Fatty Acid Synthesis in Escherichia coli: Global Positive Control by the Dual Functional Regulator FadR.J Bacteriol. 2015 Jun;197(11):1862-72. doi: 10.1128/JB.00064-15. Epub 2015 Mar 23. J Bacteriol. 2015. PMID: 25802297 Free PMC article.
References
-
- Bilder P, Lightle S, Bainbridge G, Ohren J, Finzel B, Sun F, Holley S, Al-Kassim L, Spessard C, Melnick M, Newcomer M, Waldrop GL. 2006. The structure of the carboxyltransferase component of acetyl-CoA carboxylase reveals a zinc-binding motif unique to the bacterial enzyme. Biochemistry 45:1712–1722. 10.1021/bi0520479. - DOI - PubMed
-
- Guchhait RB, Polakis SE, Dimroth P, Stoll E, Moss J, Lane MD. 1974. Acetyl coenzyme A carboxylase system of Escherichia coli. Purification and properties of the biotin carboxylase, carboxyltransferase, and carboxyl carrier protein components. J. Biol. Chem. 249:6633–6645. - PubMed
-
- Li SJ, Cronan JE., Jr 1992. The genes encoding the two carboxyltransferase subunits of Escherichia coli acetyl-CoA carboxylase. J. Biol. Chem. 267:16841–16847. - PubMed
-
- Li SJ, Cronan JE., Jr 1992. The gene encoding the biotin carboxylase subunit of Escherichia coli acetyl-CoA carboxylase. J. Biol. Chem. 267:855–863. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

