Mobilization after thrombolysis (rtPA) within 24 hours of acute stroke: what factors influence inclusion of patients in A Very Early Rehabilitation Trial (AVERT)?
- PMID: 25159047
- PMCID: PMC4236653
- DOI: 10.1186/s12883-014-0163-6
Mobilization after thrombolysis (rtPA) within 24 hours of acute stroke: what factors influence inclusion of patients in A Very Early Rehabilitation Trial (AVERT)?
Abstract
Background: A key treatment for acute ischaemic stroke is thrombolysis (rtPA). However, treatment is not devoid of side effects and patients are carefully selected. AVERT (A Very Early Rehabilitation Trial), a large, ongoing international phase III trial, tests whether starting out of bed activity within 24 hours of stroke onset improves outcome. Patients treated with rtPA can be recruited if the physician allows (447 included to date). This study aimed to identify factors that might influence the inclusion of rtPA treated patients in AVERT.
Methods: Data from all patients thrombolysed at Austin Health, Australia, between September 2007 and December 2011 were retrospectively extracted from medical records. Factors of interest included: demographic and stroke characteristics, 24 hour clinical response to rtPA treatment, cerebral imaging and process factors (day and time of admission).
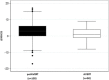
Results: 211 patients received rtPA at Austin Health and 50 (24%) were recruited to AVERT (AVERT). Of the 161 patients not recruited, 105 (65%) were eligible, and could potentially have been included (pot-AVERT). There were no significant differences in demographics, Oxfordshire classification or stroke severity (NIHSS) on admission between groups. Size and localization of stroke on imaging and symptomatic intracerebral heamorrhage rate did not differ. Patients included in AVERT showed less change in NIHSS 24 hours post rtPA (median change = 1, IQR (-1,4)) than those in the pot-AVERT group (median change = 3, IQR (0,6)) by the median difference of 2 points (95%CI:0.3; p = 0.03). A higher proportion of rtPA treated AVERT patients were admitted on weekdays (p = 0.04).
Conclusion: Excluding a possible clinical instability, no significant clinical differences were identified between thrombolysed patients included in AVERT and those who were not. Over 500 AVERT patients will be treated with rtPA at trial end. These results suggest we may be able to generalize findings to other rtPA treated patients beyond the trial population.
Figures

References
-
- Party ISW. National clinical guideline for stroke. Royal College of Physicians, London; 2012.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

