Multicomponent reactions in nucleoside chemistry
- PMID: 25161730
- PMCID: PMC4142899
- DOI: 10.3762/bjoc.10.179
Multicomponent reactions in nucleoside chemistry
Abstract
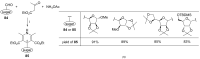
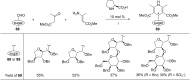
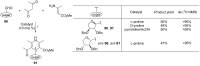
This review covers sixty original publications dealing with the application of multicomponent reactions (MCRs) in the synthesis of novel nucleoside analogs. The reported approaches were employed for modifications of the parent nucleoside core or for de novo construction of a nucleoside scaffold from non-nucleoside substrates. The cited references are grouped according to the usually recognized types of the MCRs. Biochemical properties of the novel nucleoside analogs are also presented (if provided by the authors).
Keywords: multicomponent reaction; nucleoside analog; nucleoside antibiotics; nucleoside construction; nucleoside modification.
Figures


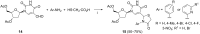
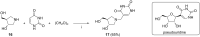
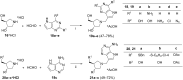
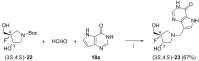






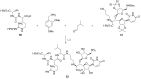

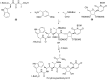
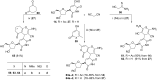

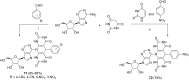


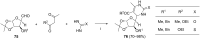





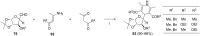


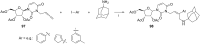

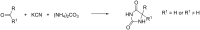
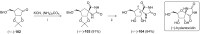
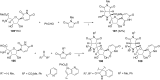

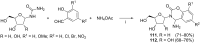
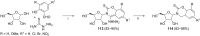
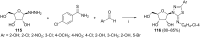
References
-
- Wiebe L I. Technetium-99m Radiopharmaceuticals: Status And Trends. Vienna: International Atomic Energy Agency; 2009. Gene and molecular imaging using 99mTc labelled radiopharmaceuticals; pp. 159–163.
-
- Alberto R, N’Dongo H P, Clericuzio M, Bonetti S, Gabano E, Cassino C, Ravera M, Osella D. Inorg Chim Acta. 2009;362:4785–4790. doi: 10.1016/j.ica.2009.06.054. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
