Decellularization of human dermis using non-denaturing anionic detergent and endonuclease: a review
- PMID: 25163609
- PMCID: PMC4426135
- DOI: 10.1007/s10561-014-9467-4
Decellularization of human dermis using non-denaturing anionic detergent and endonuclease: a review
Abstract
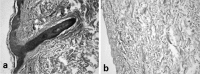
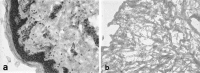
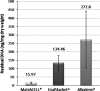
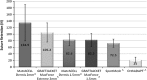
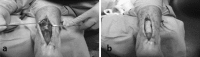
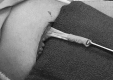
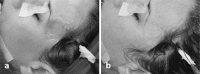
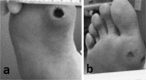
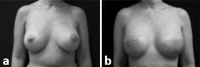
Decellularized human dermis has been used for a number of clinical applications including wound healing, soft tissue reconstruction, and sports medicine procedures. A variety of methods exist to prepare this useful class of biomaterial. Here, we describe a decellularization technology (MatrACELL(®)) utilizing a non-denaturing anionic detergent, N-Lauroyl sarcosinate, and endonuclease, which was developed to remove potentially immunogenic material while retaining biomechanical properties. Effective decellularization was demonstrated by a residual DNA content of ≤4 ng/mg of wet weight which represented >97 % DNA removal compared to unprocessed dermis. Two millimeter thick MatrACELL processed human acellular dermal matrix (MH-ADM) exhibited average ultimate tensile load to failure of 635.4 ± 199.9 N and average suture retention strength of 134.9 ± 55.1 N. Using an in vivo mouse skin excisional model, MH-ADM was shown to be biocompatible and capable of supporting cellular and vascular in-growth. Finally, clinical studies of MH-ADM in variety of applications suggest it can be an appropriate scaffold for wound healing, soft tissue reconstruction, and soft tissue augmentation.
Figures




Similar articles
-
Development and validation of cryopreserved or freeze-dried decellularized human dermis for transplantation.Cell Tissue Bank. 2024 Jun;25(2):685-695. doi: 10.1007/s10561-024-10131-6. Epub 2024 Feb 21. Cell Tissue Bank. 2024. PMID: 38381276 Free PMC article.
-
Evaluation of Detergent-Free and Detergent-Based Methods for Decellularization of Murine Skin.Tissue Eng Part A. 2018 Jun;24(11-12):955-967. doi: 10.1089/ten.TEA.2017.0273. Epub 2018 Apr 12. Tissue Eng Part A. 2018. PMID: 29303417
-
A Histological and Biomechanical Analysis of Human Acellular Dermis (HAD) Created Using a Novel Processing and Preservation Technique.Indian J Orthop. 2024 May 18;58(7):922-931. doi: 10.1007/s43465-024-01181-9. eCollection 2024 Jul. Indian J Orthop. 2024. PMID: 38948369 Free PMC article.
-
Biological incorporation of human acellular dermal matrix used in Achilles tendon repair.Cell Tissue Bank. 2017 Sep;18(3):403-411. doi: 10.1007/s10561-017-9628-3. Epub 2017 Apr 28. Cell Tissue Bank. 2017. PMID: 28455604 Free PMC article. Review.
-
Human acellular dermal wound matrix for treatment of DFU: literature review and analysis.J Wound Care. 2015 Mar;24(3):128; 129-34. doi: 10.12968/jowc.2015.24.3.128. J Wound Care. 2015. PMID: 25764957 Review.
Cited by
-
DermACELL Acellular Dermal Matrix in Oncologic Breast Reconstruction: A Cohort Study and Systematic Review.Plast Reconstr Surg Glob Open. 2022 Jun 20;10(6):e4396. doi: 10.1097/GOX.0000000000004396. eCollection 2022 Jun. Plast Reconstr Surg Glob Open. 2022. PMID: 35747252 Free PMC article.
-
Development and validation of cryopreserved or freeze-dried decellularized human dermis for transplantation.Cell Tissue Bank. 2024 Jun;25(2):685-695. doi: 10.1007/s10561-024-10131-6. Epub 2024 Feb 21. Cell Tissue Bank. 2024. PMID: 38381276 Free PMC article.
-
Glycerolized Reticular Dermis as a New Human Acellular Dermal Matrix: An Exploratory Study.PLoS One. 2016 Feb 26;11(2):e0149124. doi: 10.1371/journal.pone.0149124. eCollection 2016. PLoS One. 2016. PMID: 26918526 Free PMC article.
-
Application of benzonase in preparation of decellularized lamellar porcine corneal stroma for lamellar keratoplasty.J Biomed Mater Res A. 2019 Nov;107(11):2547-2555. doi: 10.1002/jbm.a.36760. Epub 2019 Aug 6. J Biomed Mater Res A. 2019. PMID: 31330094 Free PMC article.
-
One Step Double Augmentation with Human Dermis Allograft and Homologous PRP in Misdiagnosed and or Chronic Achilles Tendon Ruptures.Orthop Surg. 2023 Dec;15(12):3300-3308. doi: 10.1111/os.13871. Epub 2023 Sep 28. Orthop Surg. 2023. PMID: 37767601 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

