Small molecule Mcl-1 inhibitors for the treatment of cancer
- PMID: 25172548
- PMCID: PMC4340597
- DOI: 10.1016/j.pharmthera.2014.08.003
Small molecule Mcl-1 inhibitors for the treatment of cancer
Abstract
The Bcl-2 family of proteins serves as primary regulators of apoptosis. Myeloid cell leukemia 1 (Mcl-1), a pro-survival member of the Bcl-2 family of proteins, is overexpressed and the Mcl-1 gene is amplified in many tumor types. Moreover, the overexpression of Mcl-1 is the cause of resistance to several chemotherapeutic agents. Thus, Mcl-1 is a promising cancer target. This review highlights the current progress on the discovery of small molecule Mcl-1 inhibitors.
Keywords: BH3-mimetic; Inhibitors; Mcl-1; Myeloid cell leukemia-1; Small molecule.
Copyright © 2014 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures





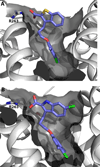
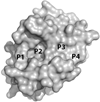
References
-
- Abulwerdi FA, Liao C, Mady A, Gavin J, Shen C, Cierpicki T, Stuckey JA, Showalter HDH, Nikolovska-Coleska Z. 3-Substituted-N-(4-Hydroxynaphthalen-1-yl)arylsulfonamides as a Novel Class of Selective Mcl-1 Inhibitors: Structure-Based Design, Synthesis, SAR and Biological Evaluation. J Med Chem. 2014;57:4111–4133. - PMC - PubMed
-
- Andersen MH, Becker JC, Straten PT. The antiapoptotic member of the Bcl-2 family Mcl-1 is a CTL target in cancer patients. Leukemia. 2005;19:484–485. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

