Glucagon-like peptide-2 regulates release of chylomicrons from the intestine
- PMID: 25173752
- PMCID: PMC4316201
- DOI: 10.1053/j.gastro.2014.08.037
Glucagon-like peptide-2 regulates release of chylomicrons from the intestine
Abstract
Background & aims: The intestine efficiently incorporates and rapidly secretes dietary fat as chylomicrons (lipoprotein particles comprising triglycerides, phospholipids, cholesterol, and proteins) that contain the apolipoprotein isoform apoB-48. The gut can store lipids for many hours after their ingestion, and release them in chylomicrons in response to oral glucose, sham feeding, or unidentified stimuli. The gut hormone glucagon-like peptide-2 (GLP-2) facilitates intestinal absorption of lipids, but its role in chylomicron secretion in human beings is unknown.
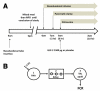
Methods: We performed a randomized, single-blind, cross-over study, with 2 study visits 4 weeks apart, to assess the effects of GLP-2 administration on triglyceride-rich lipoprotein (TRL) apoB-48 in 6 healthy men compared with placebo. Subjects underwent constant intraduodenal feeding, with a pancreatic clamp and primed constant infusion of deuterated leucine. In a separate randomized, single-blind, cross-over validation study, 6 additional healthy men ingested a high-fat meal containing retinyl palmitate and were given either GLP-2 or placebo 7 hours later with measurement of TRL triglyceride, TRL retinyl palmitate, and TRL apoB-48 levels.
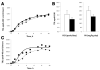
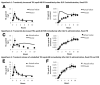
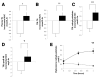
Results: GLP-2 administration resulted in a rapid (within 30 minutes) and transient increase in the concentration of TRL apoB-48, compared with placebo (P = .03). Mathematic modeling of stable isotope enrichment and the mass of the TRL apoB-48 suggested that the increase resulted from the release of stored, presynthesized apoB-48 from the gut. In the validation study, administration of GLP-2 at 7 hours after the meal, in the absence of additional food intake, robustly increased levels of TRL triglycerides (P = .007), TRL retinyl palmitate (P = .002), and TRL apoB-48 (P = .04) compared with placebo.
Conclusions: Administration of GLP-2 to men causes the release of chylomicrons that comprise previously synthesized and stored apoB-48 and lipids. This transiently increases TRL apoB-48 levels compared with placebo. Clinical trials number at www.clinicaltrials.gov: NCT 01958775.
Trial registration: ClinicalTrials.gov NCT01958775.
Keywords: Enterocyte; Fatty Acid; Human Trial; Plasma Lipid.
Copyright © 2014 AGA Institute. Published by Elsevier Inc. All rights reserved.
Figures


References
-
- Xiao C, Hsieh J, Adeli K, et al. Gut-liver interaction in triglyceride-rich lipoprotein metabolism. Am J Physiol Endocrinol Metab. 2011;301:E429–E446. - PubMed
-
- Evans K, Kuusela PJ, Cruz ML, et al. Rapid chylomicron appearance following sequential meals: effects of second meal composition. Br J Nutr. 1998;79:425–429. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

