Hirano body expression impairs spatial working memory in a novel mouse model
- PMID: 25178488
- PMCID: PMC4160558
- DOI: 10.1186/s40478-014-0131-9
Hirano body expression impairs spatial working memory in a novel mouse model
Abstract
Introduction: Hirano bodies are actin-rich intracellular inclusions found in the brains of patients with neurodegenerative conditions such as Alzheimer's disease or frontotemporal lobar degeneration-tau. While Hirano body ultrastructure and protein composition have been well studied, little is known about the physiological function of Hirano bodies in an animal model system.
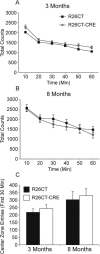
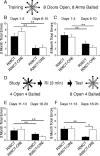
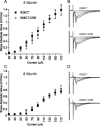
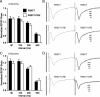
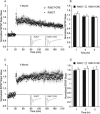
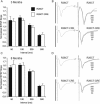
Results: Utilizing a Cre/Lox system, we have generated a new mouse model which develops an age-dependent increase in the number of model Hirano bodies present in both the CA1 region of the hippocampus and frontal cortex. These mice develop normally and experience no overt neuron loss. Mice presenting model Hirano bodies have no abnormal anxiety or locomotor activity as measured by the open field test. However, mice with model Hirano bodies develop age-dependent impairments in spatial working memory performance assessed using a delayed win-shift task in an 8-arm radial maze. Synaptic transmission, short-term plasticity, and long-term plasticity was measured in the CA1 region from slices obtained from both the ventral and dorsal hippocampus in the same mice whose spatial working memory was assessed. Baseline synaptic responses, paired pulse stimulation and long-term potentiation measurements in the ventral hippocampus were indistinguishable from control mice. In contrast, in the dorsal hippocampus, synaptic transmission at higher stimulus intensities were suppressed in 3 month old mice with Hirano bodies as compared with control mice. In addition, long-term potentiation was enhanced in the dorsal hippocampus of 8 month old mice with Hirano bodies, concurrent with observed impairment of spatial working memory. Finally, an inflammatory response was observed at 8 months of age in mice with Hirano bodies as assessed by the presence of reactive astrocytes.
Conclusion: This study shows that the presence of model Hirano bodies initiates an inflammatory response, alters hippocampal synaptic responses, and impairs spatial working memory in an age-dependent manner. This suggests that Hirano bodies may promote disease progression. This new model mouse provides a tool to investigate how Hirano bodies interact with other pathologies associated with Alzheimer's disease. Hirano bodies likely play a complex and region specific role in the brain during neurodegenerative disease progression.
Figures







Similar articles
-
Transgenic mouse model for the formation of Hirano bodies.BMC Neurosci. 2011 Oct 6;12:97. doi: 10.1186/1471-2202-12-97. BMC Neurosci. 2011. PMID: 21978358 Free PMC article.
-
Alterations in synaptic plasticity coincide with deficits in spatial working memory in presymptomatic 3xTg-AD mice.Neurobiol Learn Mem. 2015 Nov;125:152-162. doi: 10.1016/j.nlm.2015.09.003. Epub 2015 Sep 15. Neurobiol Learn Mem. 2015. PMID: 26385257 Free PMC article.
-
Enhanced long-term and impaired short-term spatial memory in GluA1 AMPA receptor subunit knockout mice: evidence for a dual-process memory model.Learn Mem. 2009 May 23;16(6):379-86. doi: 10.1101/lm.1339109. Print 2009 Jun. Learn Mem. 2009. PMID: 19470654 Free PMC article.
-
Hirano bodies and Alzheimer's disease.Kaohsiung J Med Sci. 1997 Jan;13(1):10-8. Kaohsiung J Med Sci. 1997. PMID: 9130818 Review.
-
The role of habituation in hippocampus-dependent spatial working memory tasks: evidence from GluA1 AMPA receptor subunit knockout mice.Hippocampus. 2012 May;22(5):981-94. doi: 10.1002/hipo.20896. Epub 2010 Dec 1. Hippocampus. 2012. PMID: 21125585 Free PMC article. Review.
Cited by
-
Alzheimer's disease and its co-pathologies: Implications for hippocampal degeneration, cognitive decline, and the role of APOE ε4.Alzheimers Dement. 2025 Jul;21(7):e70483. doi: 10.1002/alz.70483. Alzheimers Dement. 2025. PMID: 40665478 Free PMC article.
-
Cocaine conditioning induces persisting changes in ventral hippocampus synaptic transmission, long-term potentiation, and radial arm maze performance in the mouse.Neuropharmacology. 2019 May 15;150:27-37. doi: 10.1016/j.neuropharm.2019.02.033. Epub 2019 Mar 1. Neuropharmacology. 2019. PMID: 30831161 Free PMC article.
-
De novo actin polymerization is required for model Hirano body formation in Dictyostelium.Biol Open. 2016 Jun 15;5(6):807-18. doi: 10.1242/bio.014944. Biol Open. 2016. PMID: 27215322 Free PMC article.
-
The neuropathological diagnosis of Alzheimer's disease.Mol Neurodegener. 2019 Aug 2;14(1):32. doi: 10.1186/s13024-019-0333-5. Mol Neurodegener. 2019. PMID: 31375134 Free PMC article. Review.
-
Evaluation of delayed LNFPIII treatment initiation protocol on improving long-term behavioral and neuroinflammatory pathology in a mouse model of Gulf War Illness.Brain Behav Immun Health. 2022 Nov 8;26:100553. doi: 10.1016/j.bbih.2022.100553. eCollection 2022 Dec. Brain Behav Immun Health. 2022. PMID: 36405424 Free PMC article.
References
-
- Lynch T, Sano M, Marder KS, Bell KL, Foster NL, Defendini RF, Sima AA, Keohane C, Nygaard TG, Fahn S, Mayeux R, Rowland LP, Wilhelmensen KC. Clinical characteristics of a family with chromosome 17-linked disinhibition-demenstia-parkinsonism-amyotrophy complex. Neurol. 1994;44:1878–1884. doi: 10.1212/WNL.44.10.1878. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

