Loss of LKB1 disrupts breast epithelial cell polarity and promotes breast cancer metastasis and invasion
- PMID: 25178656
- PMCID: PMC4431490
- DOI: 10.1186/s13046-014-0070-0
Loss of LKB1 disrupts breast epithelial cell polarity and promotes breast cancer metastasis and invasion
Erratum in
-
Correction: Loss of LKB1 disrupts breast epithelial cell polarity and promotes breast cancer metastasis and invasion.J Exp Clin Cancer Res. 2025 Feb 24;44(1):66. doi: 10.1186/s13046-025-03317-7. J Exp Clin Cancer Res. 2025. PMID: 39988681 Free PMC article. No abstract available.
Abstract
Background: LKB1, also known as STK11, is a master kinase that serves as an energy metabolic sensor and is involved in cell polarity regulation. Recent studies have indicated that LKB1 is related to breast tumorigenesis and breast cancer progression. However, little work has been done on the roles of LKB1 in cell polarity and epithelial-mesenchymal transition in breast cancer. In this study, we tried to prove that loss of LKB1 disrupts breast epithelial cell polarity and causes tumor metastasis and invasion.
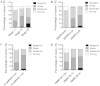
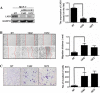
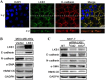
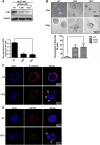
Methods: The relationships of LKB1 expression to clinic-pathological parameters and epithelial markers E-cadherin and high-molecular-weight -cytokeratin (HMW-CK) were investigated in 80 clinical breast cancer tissue samples and their paired normal control breast tissue samples by using immunohistochemistry. Then, the LKB1 expressions in metastatic and non-metastatic breast cancer cell lines were compared. The roles of LKB1 in cell polarity and epithelial-mesenchymal transition in breast cancer were determined by using immunofluorescence, western blot assay, and cell migration and invasive assays. Finally, the non-transformed human breast cell line MCF-10A was cultured in three dimensions to further reveal the role of LKB1 in breast epithelial cell polarity maintenance.
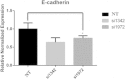
Results: Histopathological analysis showed that LKB1 expression level was significantly negatively correlated with breast cancer TNM stage, and positively correlated with ER/PR status and expression levels of E-cadherin and HMW-CK. Immunofluorescence staining showed that LKB1 was co-localized with E-cadherin at adheren junctions. In vitro analysis revealed that loss of LKB1 expression enhanced migration, invasion and the acquisition of mesenchymal phenotype, while LKB1 overexpression in MDA-MB-435 s cells, which have a low basal level of LKB1 expression, promoted the acquisition of epithelial phenotype. Finally, it was found for the first time that endogenous LKB1 knockdown resulted in abnormal cell polarity in acini formed by non-transformed breast epithelial cells grown in 3D culture.
Conclusion: Our data indicated that low expression of LKB1 was significantly associated with established markers of unfavorable breast cancer prognosis, such as loss of ER/PR, E-cadherin and HMW-CK. Knockdown of endogenous LKB1 gave rise to dysregulation of cell polarity and invasive phenotype of breast cancer cells.
Figures


References
-
- Granot Z, Swisa A, Magenheim J, Stolovich-Rain M, Fujimoto W, Manduchi E, Miki T, Lennerz JK, Stoeckert CJ Jr, Meyuhas O, Seino S, Permutt MA, Piwnica-Worms H, Bardeesy N, Dor Y. LKB1 regulates pancreatic beta cell size, polarity, and function. Cell Met. 2009;10(4):296–308. doi: 10.1016/j.cmet.2009.08.010. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

