Cannabinoid receptor type 1 antagonist, AM251, attenuates mechanical allodynia and thermal hyperalgesia after burn injury
- PMID: 25188001
- PMCID: PMC4237656
- DOI: 10.1097/ALN.0000000000000422
Cannabinoid receptor type 1 antagonist, AM251, attenuates mechanical allodynia and thermal hyperalgesia after burn injury
Abstract
Background: Burn injury causes nociceptive behaviors, and inflammation-related pathologic pain can lead to glial cell activation. This study tested the hypothesis that burn injury activates glial cells, and cannabinoid receptor 1 (CB1R) antagonist, AM251, will decrease burn pain.
Methods: Anesthetized rats received 0.75-cm third-degree burn on dorsal hind paw. Vehicle or AM251 30 μg intrathecally (older rats, n=6 per group) or, either vehicle, 0.1 or 1.0 mg/kg intraperitoneally (younger rats, n=6 per group), started immediate postburn, was administered for 7 days. Mechanical allodynia and thermal hyperalgesia were tested on ventral paw for 14 days. Microglial and astroglial activity was assessed by immunocytochemistry.
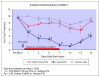
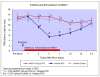
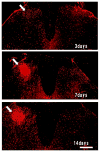
Results: Allodynia, observed on burn side from day 1 to 14, was significantly (P<0.05) attenuated by intrathecal and intraperitoneal AM251 (1 mg/kg) starting from 3 to 14 days. Hyperalgesia, observed from day 3 to 12, was completely (P<0.05) reversed by intrathecal and intraperitoneal AM251 (1 mg/kg). AM251 0.1 mg/kg had no effect. Microglial activity (n=3 per time point) increased (P<0.05) 18.5±7.5 and 12.3±1.6 (mean±SD) fold at 7 and 14 days, respectively. Astroglial activity (n=4 per time point) increased 2.9±0.3 fold at day 7 only. Glial activities were unaltered by AM251.
Conclusions: AM251 inhibited nociceptive behaviors after burn even beyond 7-day period of administration. Although many studies have documented the utility of CB1R agonists, this study indicates that endogenous cannabinoids may have an unexpected pronociceptive effect during development of burn pain, explaining why CB1R antagonist, AM251, improves nociceptive behaviors. The decreased nociception with AM251 without altering glial activity indicates that AM251 acts further downstream of activated glial cells.
Conflict of interest statement
COI: The authors declare no competing interests.
Figures




References
-
- Velayudhan L, Van Diepen E, Marudkar M, Hands O, Suribhatla S, Prettyman R, Murray J, Baillon S, Bhattacharyya S. Therapeutic potential of cannabinoids in neurodegenerative disorders: A selective review. Curr Pharm Des. 2014;20:2218–30. - PubMed
-
- Robson PJ. Therapeutic potential of cannabinoid medicines. Drug Test Anal. 2014;6:24–30. - PubMed
-
- Diezi M, Buclin T, Kuntzer T. Toxic and drug-induced peripheral neuropathies: Updates on causes, mechanisms and management. Curr Opin Neurol. 2013;26:481–8. - PubMed
-
- Zogopoulos P, Vaileiou I, Patsouris E, Theocharis SE. The role of endocannabinoids in pain modulation. Fundam Clin Pharmacol. 2013;27:64–80. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

