Divergent antiviral roles of amphibian (Xenopus laevis) macrophages elicited by colony-stimulating factor-1 and interleukin-34
- PMID: 25190077
- PMCID: PMC4226796
- DOI: 10.1189/jlb.4A0614-295R
Divergent antiviral roles of amphibian (Xenopus laevis) macrophages elicited by colony-stimulating factor-1 and interleukin-34
Abstract
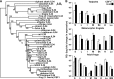
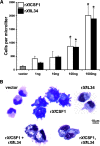
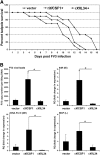
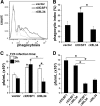
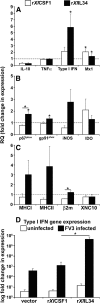
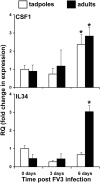
Macrophages are integral to amphibian immunity against RVs, as well as to the infection strategies of these pathogens. Although CSF-1 was considered to be the principal mediator of macrophage development, the IL-34 cytokine, which shares no sequence identity with CSF-1, is now believed to contribute to vertebrate monopoiesis. However, the respective roles of CSF-1- and IL-34-derived macrophages are still poorly understood. To delineate the contribution of these macrophage populations to amphibian immunity against the RV FV3, we identified the Xenopus laevis IL-34 and transcriptionally and functionally compared this cytokine with the previously identified X. laevis CSF-1. The X. laevis CSF-1 and IL-34 displayed strikingly nonoverlapping developmental and tissue-specific gene-expression patterns. Furthermore, only CSF-1 but not IL-34 was up-regulated in the kidneys of FV3-challenged tadpoles. Intriguingly, recombinant forms of these cytokines (rXlCSF-1, rXlIL-34) elicited morphologically distinct tadpole macrophages, and whereas rXlCSF-1 pretreatment decreased the survival of FV3-infected tadpoles, rXlIL-34 administration significantly prolonged FV3-challenged animal survival. Compared with rXlIL-34-elicited macrophages, macrophages derived by rXlCSF-1 were more phagocytic but also significantly more susceptible to in vitro FV3 infections. By contrast, rXlIL-34-derived macrophages exhibited significantly greater in vitro antiranaviral activity and displayed substantially more robust gene expression of the NADPH oxidase components (p67(phox), gp91(phox)) and type I IFN. Moreover, FV3-challenged, rXlIL-34-derived macrophages exhibited several orders of magnitude greater up-regulation of the type I IFN gene expression. This marks the first report of the disparate roles of CSF-1 and IL-34 in vertebrate antiviral immunity.
Keywords: CSF-1; FV3; IL-34; immunity; ranavirus.
© 2014 Society for Leukocyte Biology.
Figures
Similar articles
-
Amphibian macrophage development and antiviral defenses.Dev Comp Immunol. 2016 May;58:60-7. doi: 10.1016/j.dci.2015.12.008. Epub 2015 Dec 15. Dev Comp Immunol. 2016. PMID: 26705159 Free PMC article. Review.
-
Prominent amphibian (Xenopus laevis) tadpole type III interferon response to the frog virus 3 ranavirus.J Virol. 2015 May;89(9):5072-82. doi: 10.1128/JVI.00051-15. Epub 2015 Feb 25. J Virol. 2015. PMID: 25717104 Free PMC article.
-
The amphibian (Xenopus laevis) type I interferon response to frog virus 3: new insight into ranavirus pathogenicity.J Virol. 2014 May;88(10):5766-77. doi: 10.1128/JVI.00223-14. Epub 2014 Mar 12. J Virol. 2014. PMID: 24623410 Free PMC article.
-
Mechanisms of amphibian macrophage development: characterization of the Xenopus laevis colony-stimulating factor-1 receptor.Int J Dev Biol. 2014;58(10-12):757-66. doi: 10.1387/ijdb.140271jr. Int J Dev Biol. 2014. PMID: 26154317 Free PMC article.
-
Immune evasion strategies of ranaviruses and innate immune responses to these emerging pathogens.Viruses. 2012 Jul;4(7):1075-92. doi: 10.3390/v4071075. Epub 2012 Jun 28. Viruses. 2012. PMID: 22852041 Free PMC article. Review.
Cited by
-
Immunoregulatory properties of the cytokine IL-34.Cell Mol Life Sci. 2017 Jul;74(14):2569-2586. doi: 10.1007/s00018-017-2482-4. Epub 2017 Mar 3. Cell Mol Life Sci. 2017. PMID: 28258292 Free PMC article. Review.
-
The Roles of Amphibian (Xenopus laevis) Macrophages during Chronic Frog Virus 3 Infections.Viruses. 2021 Nov 18;13(11):2299. doi: 10.3390/v13112299. Viruses. 2021. PMID: 34835105 Free PMC article.
-
Amphibian macrophage development and antiviral defenses.Dev Comp Immunol. 2016 May;58:60-7. doi: 10.1016/j.dci.2015.12.008. Epub 2015 Dec 15. Dev Comp Immunol. 2016. PMID: 26705159 Free PMC article. Review.
-
Water Temperature Affects Susceptibility to Ranavirus.Ecohealth. 2016 Jun;13(2):350-9. doi: 10.1007/s10393-016-1120-1. Epub 2016 Jun 9. Ecohealth. 2016. PMID: 27283058
-
The amphibian immune system.Philos Trans R Soc Lond B Biol Sci. 2023 Jul 31;378(1882):20220123. doi: 10.1098/rstb.2022.0123. Epub 2023 Jun 12. Philos Trans R Soc Lond B Biol Sci. 2023. PMID: 37305914 Free PMC article. Review.
References
-
- Chinchar V. G. (2002) Ranaviruses (family Iridoviridae): emerging cold-blooded killers. Arch. Virol. 147, 447–470. - PubMed
-
- Williams T., Barbosa-Solomieu V., Chinchar V. G. (2005) A decade of advances in iridovirus research. Adv. Virus Res. 65, 173–248. - PubMed
-
- Chinchar V. G., Hyatt A., Miyazaki T., Williams T. (2009) Family Iridoviridae: poor viral relations no longer. Curr. Top. Microbiol. Immunol. 328, 123–170. - PubMed
-
- Bayley A. E., Hill B. J., Feist S. W. (2013) Susceptibility of the European common frog Rana temporaria to a panel of ranavirus isolates from fish and amphibian hosts. Dis. Aquat. Organ. 103, 171–183. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

