Pharmacokinetic comparison of sustained-release and standard buprenorphine in mice
- PMID: 25199095
- PMCID: PMC4113239
Pharmacokinetic comparison of sustained-release and standard buprenorphine in mice
Abstract
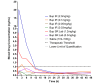
Effective pain medication is important for animal stewardship and valid research results. We compared the pharmacokinetic assessments of standard, immediate-release buprenorphine (Bup IR) and a sustained-release buprenorphine formulation (Bup SR Lab) in male C57BL/6J mice, a mouse strain commonly used in biomedical research. We postulated that the administration of Bup SR Lab would achieve a more persistent blood drug concentration (>1 ng/mL) compared with single-dose Bup IR. The study assumed a blood buprenorphine concentration of 1 ng/mL as the minimum that may result in adequate analgesia, as previously reported. The 7 experimental groups included Bup IR (0.03, 0.05, 0.1, and 2 mg/kg), Bup SR Lab (0.3 and 1.2 mg/ kg), and saline placebo (0.7 mL/100 g). Blood sampling occurred at 0.5, 1, 3, 6, 12, 24, 48, and 72 h for evaluation by using a forensic ELISA. Bup IR at 0.03 and 0.05 mg/kg and Bup SR Lab at 0.3 mg/kg failed to obtain maximal blood concentrations (Cmax) above 1 ng/mL. All other doses (0.1 and 2 mg/kg Bup IR and 1.2 mg/kg Bup SR Lab) reached a Cmax above 1 ng/mL within 3 h after injection. In addition, 1.2 mg/kg Bup SR Lab and 2 mg/kg Bup IR provided blood concentrations above 1 ng/mL for up to 12 h, and 0.1 mg/kg Bup IR achieved this criterion for as long as 3 h. In conclusion, Bup SR Lab at 1.2 mg/kg and Bup IR at 0.1 or 2.0 mg/kg achieve or surpass the published threshold for adequate analgesia in mice.
Figures


References
-
- Catbagan DL, Quimby JM, Mama KR, Rychel JK, Mich PM. 2011. Comparison of the efficacy and adverse effects of sustained-release buprenorphine hydrochloride following subcutaneous administration and buprenorphine hydrochloride following oral transmucosal administration in cats undergoing ovariohysterectomy. Am J Vet Res 72:461–466 - PubMed
-
- Chawarski MC, Schottenfeld RS, O'Connor PG, Pakes J. 1999. Plasma concentrations of buprenorphine 24 to 72 hours after dosing. Drug Alcohol Depend 55:157–163 - PubMed
-
- Christoph T, Kogel B, Schiene K, Meen M, De Vry J, Friderichs E. 2005. Broad analgesic profile of buprenorphine in rodent models of acute and chronic pain. Eur J Pharmacol 507:87–98 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
