Autocrine hemokinin-1 functions as an endogenous adjuvant for IgE-mediated mast cell inflammatory responses
- PMID: 25201259
- PMCID: PMC4362795
- DOI: 10.1016/j.jaci.2014.07.036
Autocrine hemokinin-1 functions as an endogenous adjuvant for IgE-mediated mast cell inflammatory responses
Abstract
Background: Efficient development of atopic diseases requires interactions between allergen and adjuvant to initiate and amplify the underlying inflammatory responses. Substance P (SP) and hemokinin-1 (HK-1) are neuropeptides that signal through the neurokinin-1 receptor (NK1R) to promote inflammation. Mast cells initiate the symptoms and tissue effects of atopic disorders, secreting TNF and IL-6 after FcεRI cross-linking by antigen-IgE complexes (FcεRI-activated mast cells [FcεRI-MCs]). Additionally, MCs express the NK1R, suggesting an adjuvant role for NK1R agonists in FcεRI-MC-mediated pathologies; however, in-depth research addressing this relevant aspect of MC biology is lacking.
Objective: We sought to investigate the effect of NK1R signaling and the individual roles of SP and HK-1 as potential adjuvants for FcεRI-MC-mediated allergic disorders.
Methods: Bone marrow-derived mast cells (BMMCs) from C57BL/6 wild-type (WT) or NK1R(-/-) mice were used to investigate the effects of NK1R signaling on FcεRI-MCs. BMMCs generated from Tac1(-/-) mice or after culture with Tac4 small interfering RNA were used to address the adjuvancy of SP and HK-1. WT, NK1R(-/-), and c-Kit(W-sh/W-sh) mice reconstituted with WT or NK1R(-/-) BMMCs were used to evaluate NK1R signaling on FcεRI-MC-mediated passive local and systemic anaphylaxis and on airway inflammation.
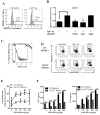
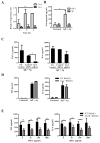
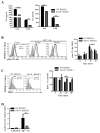
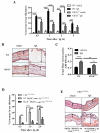
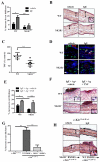
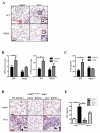
Results: FcεRI-activated MCs upregulated NK1R and HK-1 transcripts and protein synthesis, without modifying SP expression. In a positive signaling loop HK-1 promoted TNF and IL-6 secretion by MC degranulation and protein synthesis, the latter through the phosphoinositide 3-kinase/Akt/nuclear factor κB pathways. In vivo NK1R signaling was necessary for the development of passive local and systemic anaphylaxis and airway inflammation.
Conclusions: FcεRI stimulation of MCs promotes autocrine secretion of HK-1, which signals through NK1R to provide adjuvancy for efficient development of FcεRI-MC-mediated disorders.
Keywords: FcεRI; Hemokinin-1; IL-6; IgE; TNF; airway inflammation; mast cells; neurokinin-1 receptor; passive anaphylaxis; substance P.
Copyright © 2014 American Academy of Allergy, Asthma & Immunology. Published by Elsevier Inc. All rights reserved.
Figures
References
-
- Mathers AR, Tckacheva OA, Janelsins BM, Shufesky WJ, Morelli AE, Larregina AT. In vivo signaling through the neurokinin 1 receptor favors transgene expression by Langerhans cells and promotes the generation of Th1- and Tc1-biased immune responses. J Immunol. 2007;178:7006–17. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

