AhR and Arnt differentially regulate NF-κB signaling and chemokine responses in human bronchial epithelial cells
- PMID: 25201625
- PMCID: PMC4222560
- DOI: 10.1186/s12964-014-0048-8
AhR and Arnt differentially regulate NF-κB signaling and chemokine responses in human bronchial epithelial cells
Abstract
Background: The aryl hydrocarbon receptor (AhR) has gradually emerged as a regulator of inflammation in the lung and other tissues. AhR may interact with the p65-subunit of the nuclear factor (NF)-κB transcription factors, but reported outcomes of AhR/NF-κB-interactions are conflicting. Some studies suggest that AhR possess pro-inflammatory activities while others suggest that AhR may be anti-inflammatory. The present study explored the impact of AhR and its binding partner AhR nuclear translocator (Arnt) on p65-activation and two differentially regulated chemokines, CXCL8 (IL-8) and CCL5 (RANTES), in human bronchial epithelial cells (BEAS-2B).
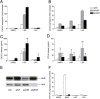
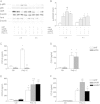
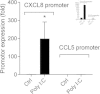
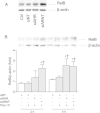
Results: Cells were exposed to CXCL8- and CCL5-inducing chemicals, 1-nitropyrene (1-NP) and 1-aminopyrene (1-AP) respectively, or the synthetic double-stranded RNA analogue, polyinosinic-polycytidylic acid (Poly I:C) which induced both chemokines. Only CXCL8, and not CCL5, appeared to be p65-dependent. Yet, constitutively active unligated AhR suppressed both CXCL8 and CCL5, as shown by siRNA knock-down and the AhR antagonist α-naphthoflavone. Moreover, AhR suppressed activation of p65 by TNF-α and Poly I:C as assessed by luciferase-assay and p65-phosphorylation at serine 536, without affecting basal p65-activity. In contrast, Arnt suppressed only CXCL8, but did not prevent the p65-activation directly. However, Arnt suppressed expression of the NF-κB-subunit RelB which is under transcriptional regulation by p65. Furthermore, AhR-ligands alone at high concentrations induced a moderate CXCL8-response, without affecting CCL5, but suppressed both CXCL8 and CCL5-responses by Poly I:C.
Conclusion: AhR and Arnt may differentially and independently regulate chemokine-responses induced by both inhaled pollutants and pulmonary infections. Constitutively active, unligated AhR suppressed the activation of p65, while Arnt may possibly interfere with the action of activated p65. Moreover, ligand-activated AhR suppressed CXCL8 and CCL5 responses by other agents, but AhR ligands alone induced CXCL8 responses when given at sufficiently high concentrations, thus underscoring the duality of AhR in regulation of inflammation. We propose that AhR-signaling may be a weak activator of p65-signaling that suppresses p65-activity induced by strong activators of NF-κB, but that its anti-inflammatory properties also are due to interference with additional pathways.
Figures





Similar articles
-
Mechanisms of chemokine responses by polycyclic aromatic hydrocarbons in bronchial epithelial cells: sensitization through toll-like receptor-3 priming.Toxicol Lett. 2013 May 23;219(2):125-32. doi: 10.1016/j.toxlet.2013.02.014. Epub 2013 Feb 28. Toxicol Lett. 2013. PMID: 23458896
-
Toll like receptor-3 priming alters diesel exhaust particle-induced cytokine responses in human bronchial epithelial cells.Toxicol Lett. 2014 Jul 3;228(1):42-7. doi: 10.1016/j.toxlet.2014.03.021. Epub 2014 Apr 4. Toxicol Lett. 2014. PMID: 24709138
-
Thrombin-induced IL-8/CXCL8 release is mediated by CK2, MSK1, and NF-κB pathways in human lung epithelial cells.Eur J Pharmacol. 2015 Nov 15;767:135-43. doi: 10.1016/j.ejphar.2015.10.018. Epub 2015 Oct 14. Eur J Pharmacol. 2015. PMID: 26463037
-
Role of AhR/ARNT system in skin homeostasis.Arch Dermatol Res. 2014 Nov;306(9):769-79. doi: 10.1007/s00403-014-1481-7. Epub 2014 Jun 26. Arch Dermatol Res. 2014. PMID: 24966027 Free PMC article. Review.
-
Regulatory crosstalk and interference between the xenobiotic and hypoxia sensing pathways at the AhR-ARNT-HIF1α signaling node.Chem Biol Interact. 2014 Jul 25;218:82-8. doi: 10.1016/j.cbi.2014.05.001. Epub 2014 May 10. Chem Biol Interact. 2014. PMID: 24824450 Free PMC article. Review.
Cited by
-
Induction of expression of aryl hydrocarbon receptor-dependent genes in human HepaRG cell line modified by shRNA and treated with β-naphthoflavone.Mol Cell Biochem. 2017 Jan;425(1-2):59-75. doi: 10.1007/s11010-016-2862-3. Epub 2016 Oct 28. Mol Cell Biochem. 2017. PMID: 27796684 Free PMC article.
-
Aryl Hydrocarbon Receptor (AhR) Limits the Inflammatory Responses in Human Lung Adenocarcinoma A549 Cells via Interference with NF-κB Signaling.Cells. 2022 Feb 17;11(4):707. doi: 10.3390/cells11040707. Cells. 2022. PMID: 35203356 Free PMC article.
-
Targeting Endothelial HIF2α/ARNT Expression for Ischemic Heart Disease Therapy.Biology (Basel). 2023 Jul 13;12(7):995. doi: 10.3390/biology12070995. Biology (Basel). 2023. PMID: 37508425 Free PMC article. Review.
-
Mononuclear phagocyte sub-types in vitro display diverse transcriptional responses to dust mite exposure.Sci Rep. 2024 Jun 20;14(1):14187. doi: 10.1038/s41598-024-64783-1. Sci Rep. 2024. PMID: 38902328 Free PMC article.
-
A delayed proinflammatory response of human preadipocytes to PCB126 is dependent on the aryl hydrocarbon receptor.Environ Sci Pollut Res Int. 2018 Jun;25(17):16481-16492. doi: 10.1007/s11356-017-9676-z. Epub 2017 Jul 11. Environ Sci Pollut Res Int. 2018. PMID: 28699004 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

