Investigating the role of uncoupling of troponin I phosphorylation from changes in myofibrillar Ca(2+)-sensitivity in the pathogenesis of cardiomyopathy
- PMID: 25202278
- PMCID: PMC4142463
- DOI: 10.3389/fphys.2014.00315
Investigating the role of uncoupling of troponin I phosphorylation from changes in myofibrillar Ca(2+)-sensitivity in the pathogenesis of cardiomyopathy
Abstract
Contraction in the mammalian heart is controlled by the intracellular Ca(2+) concentration as it is in all striated muscle, but the heart has an additional signaling system that comes into play to increase heart rate and cardiac output during exercise or stress. β-adrenergic stimulation of heart muscle cells leads to release of cyclic-AMP and the activation of protein kinase A which phosphorylates key proteins in the sarcolemma, sarcoplasmic reticulum and contractile apparatus. Troponin I (TnI) and Myosin Binding Protein C (MyBP-C) are the prime targets in the myofilaments. TnI phosphorylation lowers myofibrillar Ca(2+)-sensitivity and increases the speed of Ca(2+)-dissociation and relaxation (lusitropic effect). Recent studies have shown that this relationship between Ca(2+)-sensitivity and TnI phosphorylation may be unstable. In familial cardiomyopathies, both dilated and hypertrophic (DCM and HCM), a mutation in one of the proteins of the thin filament often results in the loss of the relationship (uncoupling) and blunting of the lusitropic response. For familial dilated cardiomyopathy in thin filament proteins it has been proposed that this uncoupling is causative of the phenotype. Uncoupling has also been found in human heart tissue from patients with hypertrophic obstructive cardiomyopathy as a secondary effect. Recently, it has been found that Ca(2+)-sensitizing drugs can promote uncoupling, whilst one Ca(2+)-desensitizing drug Epigallocatechin 3-Gallate (EGCG) can reverse uncoupling. We will discuss recent findings about the role of uncoupling in the development of cardiomyopathies and the molecular mechanism of the process.
Keywords: Ca sensitivity; cardiomyopathies; heart muscle; myofilament; phosphorylation; troponin I.
Figures
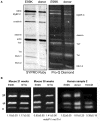
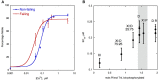

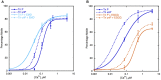
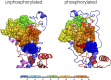
References
-
- Alves M. L., Dias F. A. L., Gaffin R. D., Simon J. N., Montminy E. M., Wolska B. M. (2014). Desensitization of myofilaments to Ca2+ as a therapeutic target for hypertrophic cardiomyopathy with mutations in thin filament proteins. Circ. Cardiovasc. Gen. 7, 132–143 10.1161/CIRCGENETICS.113.000324 - DOI - PMC - PubMed
-
- Ayaz-Guner S., Zhang J., Li L., Walker J. W., Ge Y. (2009). In vivo phosphorylation site mapping in mouse cardiac troponin i by high resolution top-down electron capture dissociation mass spectrometry: Ser22/23 are the only sites basally phosphorylated. Biochemistry 48, 8161–8170 10.1021/bi900739f - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

