Identification of the immunodominant epitope region in phospholipase A2 receptor-mediating autoantibody binding in idiopathic membranous nephropathy
- PMID: 25205735
- PMCID: PMC4310656
- DOI: 10.1681/ASN.2013121315
Identification of the immunodominant epitope region in phospholipase A2 receptor-mediating autoantibody binding in idiopathic membranous nephropathy
Abstract
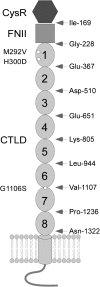
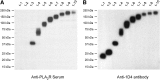
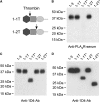
Membranous nephropathy (MN) is a common cause of nephrotic syndrome in adults. Recent clinical studies established that >70% of patients with idiopathic (also called primary) MN (IMN) possess circulating autoantibodies targeting the M-type phospholipase A2 receptor-1 (PLA2R) on the surface of glomerular visceral epithelial cells (podocytes). In situ, these autoantibodies trigger the formation of immune complexes, which are hypothesized to cause enhanced glomerular permeability to plasma proteins. Indeed, the level of autoantibody in circulation correlates with the severity of proteinuria in patients. The autoantibody only recognizes the nonreduced form of PLA2R, suggesting that disulfide bonds determine the antigenic epitope conformation. Here, we identified the immunodominant epitope region in PLA2R by probing isolated truncated PLA2R extracellular domains with sera from patients with IMN that contain anti-PLA2R autoantibodies. Patient sera specifically recognized a protein complex consisting of the cysteine-rich (CysR), fibronectin-like type II (FnII), and C-type lectin-like domain 1 (CTLD1) domains of PLA2R only under nonreducing conditions. Moreover, absence of either the CysR or CTLD1 domain prevented autoantibody recognition of the remaining domains. Additional analysis suggested that this three-domain complex contains at least one disulfide bond required for conformational configuration and autoantibody binding. Notably, the three-domain complex completely blocked the reactivity of autoantibodies from patient sera with the full-length PLA2R, and the reactivity of patient sera with the three-domain complex on immunoblots equaled the reactivity with full-length PLA2R. These results indicate that the immunodominant epitope in PLA2R is exclusively located in the CysR-FnII-CTLD1 region.
Keywords: immunology and pathology; membranous nephropathy; pathophysiology of renal disease and progression.
Copyright © 2015 by the American Society of Nephrology.
Figures







Comment in
-
The dominant humoral epitope in phospholipase A2 receptor-1: presentation matters when serving up a slice of π.J Am Soc Nephrol. 2015 Feb;26(2):237-9. doi: 10.1681/ASN.2014090877. Epub 2014 Oct 6. J Am Soc Nephrol. 2015. PMID: 25288604 Free PMC article. No abstract available.
References
-
- Glassock RJ: Pathogenesis of membranous nephropathy: A new paradigm in evolution. Contrib Nephrol 181: 131–142, 2013 - PubMed
-
- Glassock RJ: The pathogenesis of membranous nephropathy: Evolution and revolution. Curr Opin Nephrol Hypertens 21: 235–242, 2012 - PubMed
-
- Glassock RJ: Secondary membranous glomerulonephritis. Nephrol Dial Transplant 7[Suppl 1]: 64–71, 1992 - PubMed
-
- Ronco P, Debiec H: Membranous glomerulopathy: The evolving story. Curr Opin Nephrol Hypertens 19: 254–259, 2010 - PubMed
-
- Ronco P, Debiec H: Molecular pathomechanisms of membranous nephropathy: From Heymann nephritis to alloimmunization. J Am Soc Nephrol 16: 1205–1213, 2005 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

