Inorganic materials as supports for covalent enzyme immobilization: methods and mechanisms
- PMID: 25207718
- PMCID: PMC6272024
- DOI: 10.3390/molecules190914139
Inorganic materials as supports for covalent enzyme immobilization: methods and mechanisms
Abstract
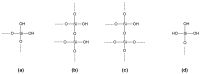
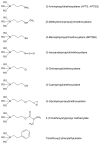
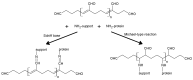
Several inorganic materials are potentially suitable for enzymatic covalent immobilization, by means of several different techniques. Such materials must meet stringent criteria to be suitable as solid matrices: complete insolubility in water, reasonable mechanical strength and chemical resistance under the operational conditions, the capability to form manageable particles with high surface area, reactivity towards derivatizing/functionalizing agents. Non-specific protein adsorption should be always considered when planning covalent immobilization on inorganic solids. A huge mass of experimental work has shown that silica, silicates, borosilicates and aluminosilicates, alumina, titania, and other oxides, are the materials of choice when attempting enzyme immobilizations on inorganic supports. More recently, some forms of elemental carbon, silicon, and certain metals have been also proposed for certain applications. With regard to the derivatization/functionalization techniques, the use of organosilanes through silanization is undoubtedly the most studied and the most applied, although inorganic bridge formation and acylation with selected acyl halides have been deeply studied. In the present article, the most common inorganic supports for covalent immobilization of the enzymes are reviewed, with particular focus on their advantages and disadvantages in terms of enzyme loadings, operational stability, undesired adsorption, and costs. Mechanisms and methods for covalent immobilization are also discussed, focusing on the most widespread activating approaches (such as glutaraldehyde, cyanogen bromide, divinylsulfone, carbodiimides, carbonyldiimidazole, sulfonyl chlorides, chlorocarbonates, N-hydroxysuccinimides).
Conflict of interest statement
The authors declare no conflict of interest.
Figures















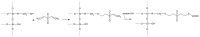









Similar articles
-
Heterofunctional supports in enzyme immobilization: from traditional immobilization protocols to opportunities in tuning enzyme properties.Biomacromolecules. 2013 Aug 12;14(8):2433-62. doi: 10.1021/bm400762h. Epub 2013 Jul 16. Biomacromolecules. 2013. PMID: 23822160 Review.
-
Immobilization of Enzymes in Protein Films.Methods Mol Biol. 2020;2100:211-226. doi: 10.1007/978-1-0716-0215-7_14. Methods Mol Biol. 2020. PMID: 31939126
-
Immobilization of Enzymes on Supports Activated with Glutaraldehyde: A Very Simple Immobilization Protocol.Methods Mol Biol. 2020;2100:119-127. doi: 10.1007/978-1-0716-0215-7_7. Methods Mol Biol. 2020. PMID: 31939119
-
Covalent immobilization of lipase onto chitosan-mesoporous silica hybrid nanomaterials by carboxyl functionalized ionic liquids as the coupling agent.Colloids Surf B Biointerfaces. 2018 May 1;165:262-269. doi: 10.1016/j.colsurfb.2018.02.033. Epub 2018 Feb 21. Colloids Surf B Biointerfaces. 2018. PMID: 29499527
-
Covalent organic frameworks as emerging host platforms for enzyme immobilization and robust biocatalysis - A review.Int J Biol Macromol. 2021 Jan 15;167:502-515. doi: 10.1016/j.ijbiomac.2020.12.002. Epub 2020 Dec 3. Int J Biol Macromol. 2021. PMID: 33279559 Review.
Cited by
-
Green Production of Cladribine by Using Immobilized 2'-Deoxyribosyltransferase from Lactobacillus delbrueckii Stabilized through a Double Covalent/Entrapment Technology.Biomolecules. 2021 Apr 29;11(5):657. doi: 10.3390/biom11050657. Biomolecules. 2021. PMID: 33947162 Free PMC article.
-
Recent advances and future perspectives of sol-gel derived porous bioactive glasses: a review.RSC Adv. 2020 Sep 11;10(56):33782-33835. doi: 10.1039/d0ra04287k. eCollection 2020 Sep 10. RSC Adv. 2020. PMID: 35519068 Free PMC article. Review.
-
An Up-to-Date Review on the Remediation of Dyes and Phenolic Compounds from Wastewaters Using Enzymes Immobilized on Emerging and Nanostructured Materials: Promises and Challenges.Nanomaterials (Basel). 2023 Jul 25;13(15):2152. doi: 10.3390/nano13152152. Nanomaterials (Basel). 2023. PMID: 37570470 Free PMC article. Review.
-
Biodegradation and biodetoxification of batik dye wastewater by laccase from Trametes hirsuta EDN 082 immobilised on light expanded clay aggregate.3 Biotech. 2021 May;11(5):247. doi: 10.1007/s13205-021-02806-8. Epub 2021 Apr 30. 3 Biotech. 2021. PMID: 33968590 Free PMC article.
-
Improved Performance of Magnetic Cross-Linked Lipase Aggregates by Interfacial Activation: A Robust and Magnetically Recyclable Biocatalyst for Transesterification of Jatropha Oil.Molecules. 2017 Dec 7;22(12):2157. doi: 10.3390/molecules22122157. Molecules. 2017. PMID: 29215562 Free PMC article.
References
-
- Minteer S.D. Methods and Protocols. A product of Humana Press; New York, NY, USA: 2011. Enzyme stabilization and immobilization. (Series: Methods in Molecular Biology).
-
- Hartmann M. Ordered mesoporous materials for bioadsorption and biocatalysis. Chem. Mater. 2005;17:4577–4593. doi: 10.1021/cm0485658. - DOI
-
- Zhao X.S., Bao X.Y., Guo W., Lee F.Y. Immobilizing catalysts on porous materials. Mater. Today. 2006;9:32–39. doi: 10.1016/S1369-7021(06)71388-8. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

