Nuclear enrichment of folate cofactors and methylenetetrahydrofolate dehydrogenase 1 (MTHFD1) protect de novo thymidylate biosynthesis during folate deficiency
- PMID: 25213861
- PMCID: PMC4207979
- DOI: 10.1074/jbc.M114.599589
Nuclear enrichment of folate cofactors and methylenetetrahydrofolate dehydrogenase 1 (MTHFD1) protect de novo thymidylate biosynthesis during folate deficiency
Abstract
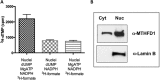
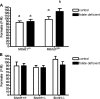
Folate-mediated one-carbon metabolism is a metabolic network of interconnected pathways that is required for the de novo synthesis of three of the four DNA bases and the remethylation of homocysteine to methionine. Previous studies have indicated that the thymidylate synthesis and homocysteine remethylation pathways compete for a limiting pool of methylenetetrahydrofolate cofactors and that thymidylate biosynthesis is preserved in folate deficiency at the expense of homocysteine remethylation, but the mechanisms are unknown. Recently, it was shown that thymidylate synthesis occurs in the nucleus, whereas homocysteine remethylation occurs in the cytosol. In this study we demonstrate that methylenetetrahydrofolate dehydrogenase 1 (MTHFD1), an enzyme that generates methylenetetrahydrofolate from formate, ATP, and NADPH, functions in the nucleus to support de novo thymidylate biosynthesis. MTHFD1 translocates to the nucleus in S-phase MCF-7 and HeLa cells. During folate deficiency mouse liver MTHFD1 levels are enriched in the nucleus >2-fold at the expense of levels in the cytosol. Furthermore, nuclear folate levels are resistant to folate depletion when total cellular folate levels are reduced by >50% in mouse liver. The enrichment of folate cofactors and MTHFD1 protein in the nucleus during folate deficiency in mouse liver and human cell lines accounts for previous metabolic studies that indicated 5,10-methylenetetrahydrofolate is preferentially directed toward de novo thymidylate biosynthesis at the expense of homocysteine remethylation during folate deficiency.
Keywords: Cell Compartmentalization; DNA Synthesis; Folate; Homocysteine; MTHFD1; Nucleoside/Nucleotide Metabolism; One-carbon Metabolism; Uracil.
© 2014 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures



Similar articles
-
Arsenic trioxide targets MTHFD1 and SUMO-dependent nuclear de novo thymidylate biosynthesis.Proc Natl Acad Sci U S A. 2017 Mar 21;114(12):E2319-E2326. doi: 10.1073/pnas.1619745114. Epub 2017 Mar 6. Proc Natl Acad Sci U S A. 2017. PMID: 28265077 Free PMC article.
-
Mthfd1 is an essential gene in mice and alters biomarkers of impaired one-carbon metabolism.J Biol Chem. 2009 Jan 16;284(3):1533-9. doi: 10.1074/jbc.M808281200. Epub 2008 Nov 25. J Biol Chem. 2009. PMID: 19033438 Free PMC article.
-
MTHFD1 regulates nuclear de novo thymidylate biosynthesis and genome stability.Biochimie. 2016 Jul;126:27-30. doi: 10.1016/j.biochi.2016.02.001. Epub 2016 Feb 4. Biochimie. 2016. PMID: 26853819 Free PMC article.
-
Cell cycle regulation of folate-mediated one-carbon metabolism.Wiley Interdiscip Rev Syst Biol Med. 2018 Nov;10(6):e1426. doi: 10.1002/wsbm.1426. Epub 2018 Jun 11. Wiley Interdiscip Rev Syst Biol Med. 2018. PMID: 29889360 Free PMC article. Review.
-
Vitamin B12-folate interrelationships.Annu Rev Nutr. 1985;5:115-41. doi: 10.1146/annurev.nu.05.070185.000555. Annu Rev Nutr. 1985. PMID: 3927946 Review.
Cited by
-
Metabolic Intermediates in Tumorigenesis and Progression.Int J Biol Sci. 2019 May 7;15(6):1187-1199. doi: 10.7150/ijbs.33496. eCollection 2019. Int J Biol Sci. 2019. PMID: 31223279 Free PMC article. Review.
-
A Simplified and Effective Approach for the Isolation of Small Pluripotent Stem Cells Derived from Human Peripheral Blood.Biomedicines. 2023 Mar 5;11(3):787. doi: 10.3390/biomedicines11030787. Biomedicines. 2023. PMID: 36979766 Free PMC article.
-
Extracellular serine and glycine are required for mouse and human skeletal muscle stem and progenitor cell function.Mol Metab. 2021 Jan;43:101106. doi: 10.1016/j.molmet.2020.101106. Epub 2020 Oct 23. Mol Metab. 2021. PMID: 33122122 Free PMC article.
-
MTHFD2 in healthy and cancer cells: Canonical and non-canonical functions.NPJ Metab Health Dis. 2024 Mar 15;2(1):3. doi: 10.1038/s44324-024-00005-6. NPJ Metab Health Dis. 2024. PMID: 40604332 Free PMC article. Review.
-
Folate nutrition and blood-brain barrier dysfunction.Curr Opin Biotechnol. 2017 Apr;44:146-152. doi: 10.1016/j.copbio.2017.01.006. Epub 2017 Feb 10. Curr Opin Biotechnol. 2017. PMID: 28189938 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

