Aging delays resolution of acute inflammation in mice: reprogramming the host response with novel nano-proresolving medicines
- PMID: 25217168
- PMCID: PMC4185223
- DOI: 10.4049/jimmunol.1401313
Aging delays resolution of acute inflammation in mice: reprogramming the host response with novel nano-proresolving medicines
Abstract
Aging is associated with an overt inflammatory phenotype and physiological decline. Specialized proresolving lipid mediators (SPMs) are endogenous autacoids that actively promote resolution of inflammation. In this study, we investigated resolution of acute inflammation in aging and the roles of SPMs. Using a self-resolving peritonitis and resolution indices coupled with lipid mediator metabololipidomics, we found that aged mice had both delayed resolution and reduced SPMs. The SPM precursor docosahexaenoic acid accelerated resolution via increased SPMs and promoted human monocyte reprogramming. In aged mice, novel nano-proresolving medicines carrying aspirin-triggered resolvins D1 and D3 reduced inflammation by promoting efferocytosis. These findings provide evidence for age-dependent resolution pathways in acute inflammation and novel means to activate resolution.
Copyright © 2014 by The American Association of Immunologists, Inc.
Conflict of interest statement
Figures


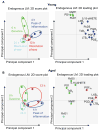
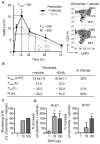




References
-
- Majno G, Joris I. Cells, Tissues, and Disease: Principles of General Pathology. Oxford University Press; New York: 2004.
-
- Gordon S, Pluddemann A. Tissue macrophage heterogeneity: issues and prospects. Semin Immunopathol. 2013;35:533–540. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

