High levels of BRC4 induced by a Tet-On 3G system suppress DNA repair and impair cell proliferation in vertebrate cells
- PMID: 25218467
- PMCID: PMC4194320
- DOI: 10.1016/j.dnarep.2014.08.003
High levels of BRC4 induced by a Tet-On 3G system suppress DNA repair and impair cell proliferation in vertebrate cells
Abstract
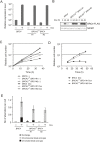
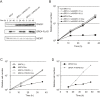
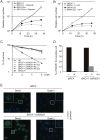
Transient induction or suppression of target genes is useful to study the function of toxic or essential genes in cells. Here we apply a Tet-On 3G system to DT40 lymphoma B cell lines, validating it for three different genes. Using this tool, we then show that overexpression of the chicken BRC4 repeat of the tumor suppressor BRCA2 impairs cell proliferation and induces chromosomal breaks. Mechanistically, high levels of BRC4 suppress double strand break-induced homologous recombination, inhibit the formation of RAD51 recombination repair foci, reduce cellular resistance to DNA damaging agents and induce a G2 damage checkpoint-mediated cell-cycle arrest. The above phenotypes are mediated by BRC4 capability to bind and inhibit RAD51. The toxicity associated with BRC4 overexpression is exacerbated by chemotherapeutic agents and reversed by RAD51 overexpression, but it is neither aggravated nor suppressed by a deficit in the non-homologous end-joining pathway of double strand break repair. We further find that the endogenous BRCA2 mediates the cytotoxicity associated with BRC4 induction, thus underscoring the possibility that BRC4 or other domains of BRCA2 cooperate with ectopic BRC4 in regulating repair activities or mitotic cell division. In all, the results demonstrate the utility of the Tet-On 3G system in DT40 research and underpin a model in which BRC4 role on cell proliferation and chromosome repair arises primarily from its suppressive role on RAD51 functions.
Keywords: BRC4; BRCA2; DT40 cells; Homologous recombination; RAD51; Tet-On system.
Copyright © 2014 The Authors. Published by Elsevier B.V. All rights reserved.
Figures

References
-
- Venkitaraman A.R. Chromosome stability, DNA recombination and the BRCA2 tumour suppressor. Curr. Opin. Cell Biol. 2001;13:338–343. - PubMed
-
- Wong A.K., Pero R., Ormonde P.A., Tavtigian S.V., Bartel P.L. RAD51 interacts with the evolutionarily conserved BRC motifs in the human breast cancer susceptibility gene BRCA2. J. Biol. Chem. 1997;272:31941–31944. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

