Axon plasticity in the mammalian central nervous system after injury
- PMID: 25218468
- PMCID: PMC4190162
- DOI: 10.1016/j.tins.2014.08.008
Axon plasticity in the mammalian central nervous system after injury
Abstract
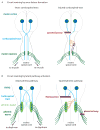
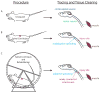
It is widely recognized that severed axons in the adult central nervous system (CNS) have limited capacity to regenerate. However, mounting evidence from studies of CNS injury response and repair is challenging the prevalent view that the adult mammalian CNS is incapable of structural reorganization to adapt to an altered environment. Animal studies demonstrate the potential to achieve significant anatomical repair and functional recovery following CNS injury by manipulating axon growth regulators alone or in combination with activity-dependent strategies. With a growing understanding of the cellular and molecular mechanisms regulating axon plasticity, and the availability of new experimental tools to map detour circuits of functional importance, directing circuit rewiring to promote functional recovery may be achieved.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures
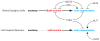
Similar articles
-
Concept and molecular basis of axonal regeneration after central nervous system injury.Neurosci Res. 2014 Jan;78:45-9. doi: 10.1016/j.neures.2013.07.002. Epub 2013 Jul 26. Neurosci Res. 2014. PMID: 23896200 Review.
-
Gene-Silencing Screen for Mammalian Axon Regeneration Identifies Inpp5f (Sac2) as an Endogenous Suppressor of Repair after Spinal Cord Injury.J Neurosci. 2015 Jul 22;35(29):10429-39. doi: 10.1523/JNEUROSCI.1718-15.2015. J Neurosci. 2015. PMID: 26203138 Free PMC article.
-
Sulfated glycans in network rewiring and plasticity after neuronal injuries.Neurosci Res. 2014 Jan;78:50-4. doi: 10.1016/j.neures.2013.10.005. Epub 2013 Oct 21. Neurosci Res. 2014. PMID: 24157431 Review.
-
Proteoglycans in the central nervous system: plasticity, regeneration and their stimulation with chondroitinase ABC.Restor Neurol Neurosci. 2008;26(2-3):131-45. Restor Neurol Neurosci. 2008. PMID: 18820407 Review.
-
Axon growth and synaptic function: A balancing act for axonal regeneration and neuronal circuit formation in CNS trauma and disease.Dev Neurobiol. 2020 Jul;80(7-8):277-301. doi: 10.1002/dneu.22780. Epub 2020 Sep 28. Dev Neurobiol. 2020. PMID: 32902152 Free PMC article. Review.
Cited by
-
Oligodendrocyte involvement in Gulf War Illness.Glia. 2019 Nov;67(11):2107-2124. doi: 10.1002/glia.23668. Epub 2019 Jul 24. Glia. 2019. PMID: 31339622 Free PMC article. Review.
-
Neuropilin-1-mediated pruning of corticospinal tract fibers is required for motor recovery after spinal cord injury.Cell Death Dis. 2019 Jan 25;10(2):67. doi: 10.1038/s41419-019-1338-2. Cell Death Dis. 2019. PMID: 30683854 Free PMC article.
-
Magnetic Field Promotes Migration of Schwann Cells with Chondroitinase ABC (ChABC)-Loaded Superparamagnetic Nanoparticles Across Astrocyte Boundary in vitro.Int J Nanomedicine. 2020 Jan 20;15:315-332. doi: 10.2147/IJN.S227328. eCollection 2020. Int J Nanomedicine. 2020. PMID: 32021182 Free PMC article.
-
Co-occupancy identifies transcription factor co-operation for axon growth.Nat Commun. 2021 May 5;12(1):2555. doi: 10.1038/s41467-021-22828-3. Nat Commun. 2021. PMID: 33953205 Free PMC article.
-
A surviving intact branch stabilizes remaining axon architecture after injury as revealed by in vivo imaging in the mouse spinal cord.Neuron. 2015 May 20;86(4):947-954. doi: 10.1016/j.neuron.2015.03.061. Epub 2015 Apr 30. Neuron. 2015. PMID: 25937174 Free PMC article.
References
-
- David S, Aguayo AJ. Axonal elongation into peripheral nervous system “bridges” after central nervous system injury in adult rats. Science. 1981;214:931–933. - PubMed
-
- Oudega M, et al. Regeneration of adult rat sensory axons into intraspinal nerve grafts: promoting effects of conditioning lesion and graft predegeneration. Experimental neurology. 1994;129:194–206. - PubMed
-
- Richardson PM, Issa VM. Peripheral injury enhances central regeneration of primary sensory neurones. Nature. 1984;309:791–793. - PubMed
-
- Goldberg JL, et al. Amacrine-signaled loss of intrinsic axon growth ability by retinal ganglion cells. Science. 2002;296:1860–1864. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

