UniChem: extension of InChI-based compound mapping to salt, connectivity and stereochemistry layers
- PMID: 25221628
- PMCID: PMC4158273
- DOI: 10.1186/s13321-014-0043-5
UniChem: extension of InChI-based compound mapping to salt, connectivity and stereochemistry layers
Abstract
UniChem is a low-maintenance, fast and freely available compound identifier mapping service, recently made available on the Internet. Until now, the criterion of molecular equivalence within UniChem has been on the basis of complete identity between Standard InChIs. However, a limitation of this approach is that stereoisomers, isotopes and salts of otherwise identical molecules are not considered as related. Here, we describe how we have exploited the layered structural representation of the Standard InChI to create new functionality within UniChem that integrates these related molecular forms. The service, called 'Connectivity Search' allows molecules to be first matched on the basis of complete identity between the connectivity layer of their corresponding Standard InChIs, and the remaining layers then compared to highlight stereochemical and isotopic differences. Parsing of Standard InChI sub-layers permits mixtures and salts to also be included in this integration process. Implementation of these enhancements required simple modifications to the schema, loader and web application, but none of which have changed the original UniChem functionality or services. The scope of queries may be varied using a variety of easily configurable options, and the output is annotated to assist the user to filter, sort and understand the difference between query and retrieved structures. A RESTful web service output may be easily processed programmatically to allow developers to present the data in whatever form they believe their users will require, or to define their own level of molecular equivalence for their resource, albeit within the constraint of identical connectivity.
Keywords: Chemical databases; Connectivity search; Data integration; InChIKey; Standard InChI; UniChem.
Figures

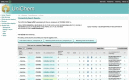

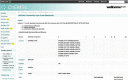
References
-
- NIH Chemical Identifier Resolver., [http://cactus.nci.nih.gov/chemical/structure]
-
- ChemSpider., [http://www.chemspider.com/]
-
- Williams A, Tkachenko V: The Royal Society of Chemistry and the delivery of chemistry data repositories for the community.J Comput Aided Mol Des 2014., - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

