Diverse functional evolution of serine decarboxylases: identification of two novel acetaldehyde synthases that uses hydrophobic amino acids as substrates
- PMID: 25230835
- PMCID: PMC4177580
- DOI: 10.1186/s12870-014-0247-x
Diverse functional evolution of serine decarboxylases: identification of two novel acetaldehyde synthases that uses hydrophobic amino acids as substrates
Abstract
Background: Type II pyridoxal 5'-phosphate decarboxylases are an important group of phylogenetically diverse enzymes involved in amino acid metabolism. Within plants, this group of enzymes is represented by aromatic amino acid decarboxylases, glutamate decarboxylases and serine decarboxylases. Additional evolutionary divergence of plant aromatic amino acid decarboxylases has resulted in further subcategories with distinct substrate specificities and enzymatic activities. Despite shared homology, no such evolutionary divergence has been characterized within glutamate decarboxylases or serine decarboxylases (SDC).
Results: Comparative analysis of two previously characterized serine decarboxylase-like (SDC-like) enzymes demonstrates distinct substrate specificities despite their highly conserved primary sequence. The alternate substrate preference of these homologous SDC-like proteins indicated that functional divergence might have occurred with in SDC-like proteins. In an effort to identify additional SDC-like functional divergence, two uncharacterized SDC-like enzymes were recombinantly expressed and characterized.
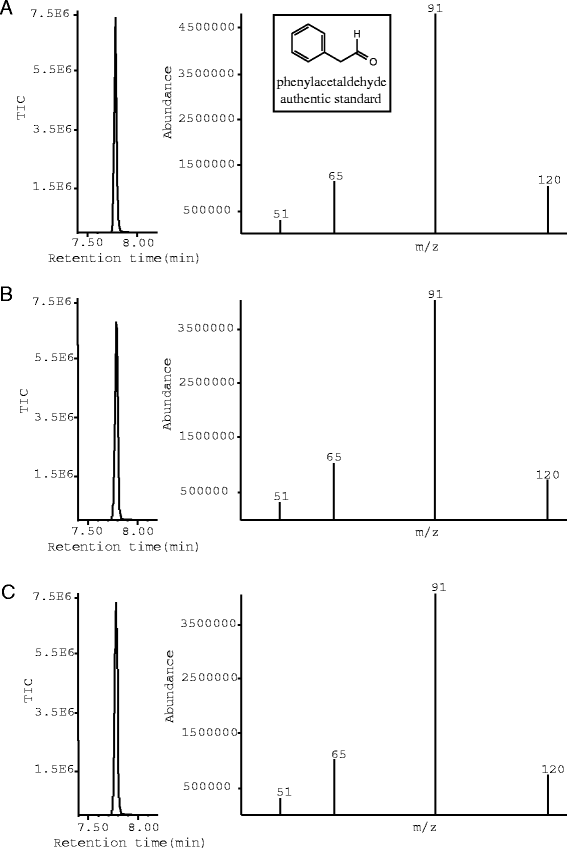
Conclusions: An extensive biochemical analysis of two serine decarboxylases-like recombinant proteins led to an interesting discovery; both proteins catalyze the formation of acetaldehyde derivatives from select hydrophobic amino acids substrates. Specifically, Medicago truncatula [GenBank: XP_003592128] and Cicer arietinum [GenBank: XP_004496485] catalyze the decarboxylation and oxidative deamination of phenylalanine, methionine, leucine and tryptophan to generate their corresponding acetaldehydes. The promiscuous aldehyde synthase activity of these proteins yields novel products of 4-(methylthio) butanal, 3-methylbutanal (isovaleraldehyde) and indole-3-acetaldehyde from methionine, leucine and tryptophan respectively. A comparative biochemical analysis of the Medicago truncatula and Cicer arietinum enzymes against two previously characterized SDC-like enzymes further emphasizes the unusual substrate specificity and activity of these novel aldehyde synthases. Due to the strong substrate preference towards phenylalanine, it is likely that both enzymes function as phenylacetaldehyde synthesis in vivo. However, due to their significant sequence divergence and unusual substrate promiscuity these enzymes are functionally and evolutionary divergent from canonical phenylacetaldehyde synthesis enzymes. This work further elaborates on the functional complexity of plant type II PLP decarboxylases and their roles in secondary metabolite biosynthesis.
Figures



Similar articles
-
Structural basis for divergent and convergent evolution of catalytic machineries in plant aromatic amino acid decarboxylase proteins.Proc Natl Acad Sci U S A. 2020 May 19;117(20):10806-10817. doi: 10.1073/pnas.1920097117. Epub 2020 May 5. Proc Natl Acad Sci U S A. 2020. PMID: 32371491 Free PMC article.
-
Biochemical evaluation of the decarboxylation and decarboxylation-deamination activities of plant aromatic amino acid decarboxylases.J Biol Chem. 2013 Jan 25;288(4):2376-87. doi: 10.1074/jbc.M112.401752. Epub 2012 Nov 30. J Biol Chem. 2013. PMID: 23204519 Free PMC article.
-
Multiple evolutionary origin of pyridoxal-5'-phosphate-dependent amino acid decarboxylases.Eur J Biochem. 1994 May 1;221(3):997-1002. doi: 10.1111/j.1432-1033.1994.tb18816.x. Eur J Biochem. 1994. PMID: 8181483
-
Plant aromatic L-amino acid decarboxylases: evolution, biochemistry, regulation, and metabolic engineering applications.Phytochemistry. 2000 May;54(2):121-38. doi: 10.1016/s0031-9422(00)00050-9. Phytochemistry. 2000. PMID: 10872203 Review.
-
New Insights Emerging from Recent Investigations on Human Group II Pyridoxal 5'-Phosphate Decarboxylases.Curr Med Chem. 2017;24(3):226-244. doi: 10.2174/0929867324666161123093339. Curr Med Chem. 2017. PMID: 27881066 Review.
Cited by
-
Structural Basis of the Substrate Specificity and Enzyme Catalysis of a Papaver somniferum Tyrosine Decarboxylase.Front Mol Biosci. 2017 Feb 9;4:5. doi: 10.3389/fmolb.2017.00005. eCollection 2017. Front Mol Biosci. 2017. Retraction in: Front Mol Biosci. 2017 Sep 27;4:69. doi: 10.3389/fmolb.2017.00069. PMID: 28232911 Free PMC article. Retracted.
-
Current Advances on Structure-Function Relationships of Pyridoxal 5'-Phosphate-Dependent Enzymes.Front Mol Biosci. 2019 Mar 5;6:4. doi: 10.3389/fmolb.2019.00004. eCollection 2019. Front Mol Biosci. 2019. PMID: 30891451 Free PMC article. Review.
-
Branched-Chain Volatiles in Fruit: A Molecular Perspective.Front Plant Sci. 2022 Jan 27;12:814138. doi: 10.3389/fpls.2021.814138. eCollection 2021. Front Plant Sci. 2022. PMID: 35154212 Free PMC article. Review.
-
Aromatic amino acid decarboxylase is involved in volatile phenylacetaldehyde production in loquat (Eriobotrya japonica) flowers.Plant Biotechnol (Tokyo). 2017;34(4):193-198. doi: 10.5511/plantbiotechnology.17.0926a. Epub 2017 Dec 2. Plant Biotechnol (Tokyo). 2017. PMID: 31275027 Free PMC article.
-
Structural basis for divergent and convergent evolution of catalytic machineries in plant aromatic amino acid decarboxylase proteins.Proc Natl Acad Sci U S A. 2020 May 19;117(20):10806-10817. doi: 10.1073/pnas.1920097117. Epub 2020 May 5. Proc Natl Acad Sci U S A. 2020. PMID: 32371491 Free PMC article.
References
-
- Rhodes D, Hanson AD. Quaternary ammonium and tertiary sulfonium compounds in higher plants. Annu Rev Plant Physiol Plant Mol Biol. 1993;44:357–384. doi: 10.1146/annurev.pp.44.060193.002041. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

