Graphene oxide-based SPR biosensor chip for immunoassay applications
- PMID: 25232298
- PMCID: PMC4154937
- DOI: 10.1186/1556-276X-9-445
Graphene oxide-based SPR biosensor chip for immunoassay applications
Abstract
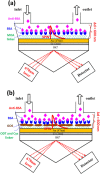
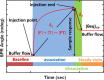
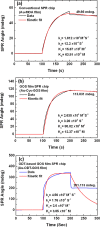
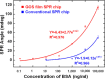
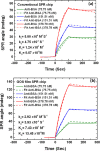
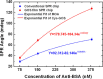
This work develops a highly sensitive immunoassay sensor for use in graphene oxide sheet (GOS)-based surface plasmon resonance (SPR) chips. This sensing film, which is formed by chemically modifying a GOS surface, has covalent bonds that strongly interact with the bovine serum albumin (BSA), explaining why it has a higher sensitivity. This GOS film-based SPR chip has a BSA concentration detection limit that is 100 times higher than that of the conventional Au-film-based sensor. The affinity constants (K A) on the GOS film-based SPR chip and the conventional SPR chip for 100 μg/ml BSA are 80.82 × 10(6) M(-1) and 15.67 × 10(6) M(-1), respectively. Therefore, the affinity constant of the GOS film-based SPR chip is 5.2 times higher than that of the conventional chip. With respect to the protein-protein interaction, the SPR sensor capability to detect angle changes at a low concentration anti-BSA of 75.75 nM on the GOS film-based SPR chip and the conventional SPR chip is 36.1867 and 26.1759 mdeg, respectively. At a high concentration, anti-BSA of 378.78 nM on the GOS film-based SPR chip and the conventional SPR chip reveals two times increases in the SPR angle shift. Above results demonstrate that the GOS film is promising for highly sensitive clinical diagnostic applications.
Keywords: Bovine albumin serum (BSA); Graphene oxide sheet (GOS); Protein; Surface plasmon resonance (SPR).
Figures



Similar articles
-
Exploring Graphene and MoS2 Chips Based Surface Plasmon Resonance Biosensors for Diagnostic Applications.Front Chem. 2020 Aug 26;8:728. doi: 10.3389/fchem.2020.00728. eCollection 2020. Front Chem. 2020. PMID: 33005604 Free PMC article. Review.
-
Carboxyl-functionalized graphene oxide composites as SPR biosensors with enhanced sensitivity for immunoaffinity detection.Biosens Bioelectron. 2017 Mar 15;89(Pt 1):370-376. doi: 10.1016/j.bios.2016.06.073. Epub 2016 Jun 25. Biosens Bioelectron. 2017. PMID: 27396822
-
Affinity capture surface carboxyl-functionalized MoS2 sheets to enhance the sensitivity of surface plasmon resonance immunosensors.Talanta. 2018 Aug 1;185:174-181. doi: 10.1016/j.talanta.2018.03.073. Epub 2018 Mar 27. Talanta. 2018. PMID: 29759186
-
Highly sensitive and selective surface plasmon resonance sensor for detection of sub-ppb levels of benzo[a]pyrene by indirect competitive immunoreaction method.Biosens Bioelectron. 2003 Jul;18(7):953-9. doi: 10.1016/s0956-5663(02)00242-7. Biosens Bioelectron. 2003. PMID: 12713919
-
Recent advances in the development of graphene-based surface plasmon resonance (SPR) interfaces.Anal Bioanal Chem. 2013 Feb;405(5):1435-43. doi: 10.1007/s00216-012-6624-0. Epub 2013 Jan 15. Anal Bioanal Chem. 2013. PMID: 23314618 Review.
Cited by
-
Exploring Graphene and MoS2 Chips Based Surface Plasmon Resonance Biosensors for Diagnostic Applications.Front Chem. 2020 Aug 26;8:728. doi: 10.3389/fchem.2020.00728. eCollection 2020. Front Chem. 2020. PMID: 33005604 Free PMC article. Review.
-
Surface Plasmon Resonance Biosensors with Magnetic Sandwich Hybrids for Signal Amplification.Biosensors (Basel). 2022 Jul 22;12(8):554. doi: 10.3390/bios12080554. Biosensors (Basel). 2022. PMID: 35892451 Free PMC article.
-
Clinical Application of a Graphene Oxide-Based Surface Plasmon Resonance Biosensor to Measure First-Trimester Serum Pregnancy-Associated Plasma Protein-A/A2 Ratio to Predict Preeclampsia.Int J Nanomedicine. 2023 Dec 7;18:7469-7481. doi: 10.2147/IJN.S438426. eCollection 2023. Int J Nanomedicine. 2023. PMID: 38090367 Free PMC article.
-
Enhanced Plasmonic Biosensors of Hybrid Gold Nanoparticle-Graphene Oxide-Based Label-Free Immunoassay.Nanoscale Res Lett. 2018 May 16;13(1):152. doi: 10.1186/s11671-018-2565-7. Nanoscale Res Lett. 2018. PMID: 29767347 Free PMC article.
-
Label-free plasmonic-based biosensing using a gold nanohole array chip coated with a wafer-scale deposited WS2 monolayer.RSC Adv. 2022 Nov 21;12(51):33284-33292. doi: 10.1039/d2ra03479d. eCollection 2022 Nov 15. RSC Adv. 2022. PMID: 36425154 Free PMC article.
References
-
- Yan H, Low T, Zhu W, Wu Y, Freitag M, Li X, Guinea F, Avouris P, Xia F. Damping pathways of mid-infrared plasmons in graphene nanostructures. Nat Photon. 2013;9:394–399. doi: 10.1038/nphoton.2013.57. - DOI
-
- Jablan M, Soljacic M, Buljan H. Plasmons in graphene: fundamental properties and potential applications. Proc IEEE. 2013;9:1689.
LinkOut - more resources
Full Text Sources
Other Literature Sources

