Negative feedback regulation of Wnt signaling via N-linked fucosylation in zebrafish
- PMID: 25238963
- PMCID: PMC4259045
- DOI: 10.1016/j.ydbio.2014.09.010
Negative feedback regulation of Wnt signaling via N-linked fucosylation in zebrafish
Abstract
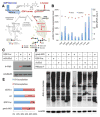
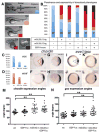
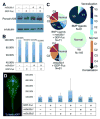
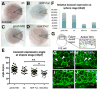
L-fucose, a monosaccharide widely distributed in eukaryotes and certain bacteria, is a determinant of many functional glycans that play central roles in numerous biological processes. The molecular mechanism, however, by which fucosylation mediates these processes remains largely elusive. To study how changes in fucosylation impact embryonic development, we up-regulated N-linked fucosylation via over-expression of a key GDP-Fucose transporter, Slc35c1, in zebrafish. We show that Slc35c1 overexpression causes elevated N-linked fucosylation and disrupts embryonic patterning in a transporter activity dependent manner. We demonstrate that patterning defects associated with enhanced N-linked fucosylation are due to diminished canonical Wnt signaling. Chimeric analyses demonstrate that elevated Slc35c1 expression in receiving cells decreases the signaling range of Wnt8a during zebrafish embryogenesis. Moreover, we provide biochemical evidence that this decrease is associated with reduced Wnt8 ligand and elevated Lrp6 coreceptor, which we show are both substrates for N-linked fucosylation in zebrafish embryos. Strikingly, slc35c1 expression is regulated by canonical Wnt signaling. These results suggest that Wnt limits its own signaling activity in part via up-regulation of a transporter, slc35c1 that promotes terminal fucosylation and thereby limits Wnt activity.
Keywords: Fucosylation; GDP-Fucose transporter; Wnt signaling; Zebrafish patterning; slc35c1.
Copyright © 2014 Elsevier Inc. All rights reserved.
Conflict of interest statement
All the authors agreed to the manuscript’s contents. The authors do not have any financial conflict of interest that might be construed to influence the results or interpretation of the manuscript.
Figures

Similar articles
-
In Situ Fucosylation of the Wnt Co-receptor LRP6 Increases Its Endocytosis and Reduces Wnt/β-Catenin Signaling.Cell Chem Biol. 2020 Sep 17;27(9):1140-1150.e4. doi: 10.1016/j.chembiol.2020.06.015. Epub 2020 Jul 9. Cell Chem Biol. 2020. PMID: 32649905 Free PMC article.
-
Slc35c2 promotes Notch1 fucosylation and is required for optimal Notch signaling in mammalian cells.J Biol Chem. 2010 Nov 12;285(46):36245-54. doi: 10.1074/jbc.M110.126003. Epub 2010 Sep 13. J Biol Chem. 2010. PMID: 20837470 Free PMC article.
-
Incorporation of fucose into glycans independent of the GDP-fucose transporter SLC35C1 preferentially utilizes salvaged over de novo GDP-fucose.J Biol Chem. 2022 Aug;298(8):102206. doi: 10.1016/j.jbc.2022.102206. Epub 2022 Jun 27. J Biol Chem. 2022. PMID: 35772493 Free PMC article.
-
Fucosylation in cancer biology and its clinical applications.Prog Mol Biol Transl Sci. 2019;162:93-119. doi: 10.1016/bs.pmbts.2019.01.002. Epub 2019 Mar 6. Prog Mol Biol Transl Sci. 2019. PMID: 30905466 Review.
-
Wnt signaling in vertebrate axis specification.Cold Spring Harb Perspect Biol. 2013 Jan 1;5(1):a007955. doi: 10.1101/cshperspect.a007955. Cold Spring Harb Perspect Biol. 2013. PMID: 22914799 Free PMC article. Review.
Cited by
-
CDG Therapies: From Bench to Bedside.Int J Mol Sci. 2018 Apr 27;19(5):1304. doi: 10.3390/ijms19051304. Int J Mol Sci. 2018. PMID: 29702557 Free PMC article. Review.
-
Modulating Cell-Surface Receptor Signaling and Ion Channel Functions by In Situ Glycan Editing.Angew Chem Int Ed Engl. 2018 Jan 22;57(4):967-971. doi: 10.1002/anie.201706535. Epub 2018 Jan 2. Angew Chem Int Ed Engl. 2018. PMID: 29292859 Free PMC article.
-
Down-regulation of SLC35C1 induces colon cancer through over-activating Wnt pathway.J Cell Mol Med. 2020 Mar;24(5):3079-3090. doi: 10.1111/jcmm.14969. Epub 2020 Jan 21. J Cell Mol Med. 2020. PMID: 31961998 Free PMC article.
-
Small-Molecule-Mediated Suppression of BMP Signaling by Selective Inhibition of BMP1-Dependent Chordin Cleavage.Int J Mol Sci. 2023 Feb 21;24(5):4313. doi: 10.3390/ijms24054313. Int J Mol Sci. 2023. PMID: 36901744 Free PMC article.
-
Targeting the NOTCH2/ADAM10/TCF7L2 Axis-Mediated Transcriptional Regulation of Wnt Pathway Suppresses Tumor Growth and Enhances Chemosensitivity in Colorectal Cancer.Adv Sci (Weinh). 2025 Jan;12(3):e2405758. doi: 10.1002/advs.202405758. Epub 2024 Nov 27. Adv Sci (Weinh). 2025. PMID: 39601111 Free PMC article.
References
-
- Becker DJ, Lowe JB. Fucose: biosynthesis and biological function in mammals. Glycobiology. 2003;13:41R–53R. - PubMed
-
- Cheyette BN, Waxman JS, Miller JR, Takemaru K, Sheldahl LC, Khlebtsova N, Fox EP, Earnest T, Moon RT. Dapper, a Dishevelled-associated antagonist of beta-catenin and JNK signaling, is required for notochord formation. Dev Cell. 2002;2:449–461. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

