Nevirapine or efavirenz for tuberculosis and HIV coinfected patients: exposure and virological failure relationship
- PMID: 25239466
- PMCID: PMC4267502
- DOI: 10.1093/jac/dku348
Nevirapine or efavirenz for tuberculosis and HIV coinfected patients: exposure and virological failure relationship
Abstract
Objectives: We describe nevirapine and efavirenz exposure on and off tuberculosis treatment and consequences for virological efficacy and tolerance in patients included in the ANRS 12146/12214-CARINEMO trial.
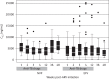
Methods: Participants were randomly selected to receive either nevirapine at 200 mg twice daily (n = 256) or efavirenz at 600 mg daily (n = 270), both combined with two nucleoside analogues. Blood samples were drawn 12 h after nevirapine or efavirenz administration, while on tuberculosis treatment and after tuberculosis treatment discontinuation. In 62 participants, samples taken 12 h after drug administration were drawn weekly for the first month of ART. Sixteen participants participated in an extensive pharmacokinetic study of nevirapine. Concentrations were compared with the therapeutic ranges of 3000-8000 ng/mL for nevirapine and 1000-4000 ng/mL for efavirenz.
Results: Nevirapine concentrations at the end of the first week of treatment (on antituberculosis drugs) did not differ from concentrations off tuberculosis treatment, but declined thereafter. Concentrations at steady-state were 4111 ng/mL at week 12 versus 6095 ng/mL at week 48 (P < 0.0001). Nevirapine concentrations <3000 ng/mL were found to be a risk factor for virological failure. Efavirenz concentrations were higher on than off tuberculosis treatment (2700 versus 2450 ng/mL, P < 0.0001).
Conclusions: The omission of the 2 week lead-in dose of nevirapine prevented low concentrations at treatment initiation but did not prevent the risk of virological failure. Results support the WHO recommendation to use efavirenz at 600 mg daily in patients on rifampicin-based antituberculosis therapy.
Keywords: TB/HIV coinfection; drug–drug interactions; efavirenz; nevirapine.
© The Author 2014. Published by Oxford University Press on behalf of the British Society for Antimicrobial Chemotherapy.
Figures
References
-
- Abdool Karim Q, Abdool Karim SS, Baxter C, et al. The SAPIT trial provides essential evidence on risks and benefits of integrated and sequential treatment of HIV and tuberculosis. S Afr Med J. 2010;100:808–9. - PubMed
-
- WHO. Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection: Recommendations for a Public Health Approach. June 2013. http://www.who.int/hiv/pub/guidelines/arv2013/download/en/index.html . - PubMed
-
- Scourfield A, Zheng J, Chinthapalli S, et al. Discontinuation of Atripla as first-line therapy in HIV-1 infected individuals. AIDS. 2012;26:1399–401. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

