Interrogating two schedules of the AKT inhibitor MK-2206 in patients with advanced solid tumors incorporating novel pharmacodynamic and functional imaging biomarkers
- PMID: 25239610
- PMCID: PMC4233149
- DOI: 10.1158/1078-0432.CCR-14-0868
Interrogating two schedules of the AKT inhibitor MK-2206 in patients with advanced solid tumors incorporating novel pharmacodynamic and functional imaging biomarkers
Abstract
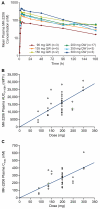
Purpose: Multiple cancers harbor genetic aberrations that impact AKT signaling. MK-2206 is a potent pan-AKT inhibitor with a maximum tolerated dose (MTD) previously established at 60 mg on alternate days (QOD). Due to a long half-life (60-80 hours), a weekly (QW) MK-2206 schedule was pursued to compare intermittent QW and continuous QOD dosing.
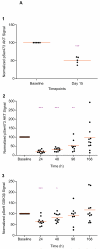
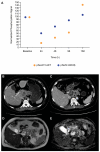
Experimental design: Patients with advanced cancers were enrolled in a QW dose-escalation phase I study to investigate the safety and pharmacokinetic-pharmacodynamic profiles of tumor and platelet-rich plasma (PRP). The QOD MTD of MK-2206 was also assessed in patients with ovarian and castration-resistant prostate cancers and patients with advanced cancers undergoing multiparametric functional magnetic resonance imaging (MRI) studies, including dynamic contrast-enhanced MRI, diffusion-weighted imaging, magnetic resonance spectroscopy, and intrinsic susceptibility-weighted MRI.
Results: A total of 71 patients were enrolled; 38 patients had 60 mg MK-2206 QOD, whereas 33 received MK-2206 at 90, 135, 150, 200, 250, and 300 mg QW. The QW MK-2206 MTD was established at 200 mg following dose-limiting rash at 250 and 300 mg. QW dosing appeared to be similarly tolerated to QOD, with toxicities including rash, gastrointestinal symptoms, fatigue, and hyperglycemia. Significant AKT pathway blockade was observed with both continuous QOD and intermittent QW dosing of MK-2206 in serially obtained tumor and PRP specimens. The functional imaging studies demonstrated that complex multiparametric MRI protocols may be effectively implemented in a phase I trial.
Conclusions: Treatment with MK-2206 safely results in significant AKT pathway blockade in QOD and QW schedules. The intermittent dose of 200 mg QW is currently used in phase II MK-2206 monotherapy and combination studies (NCT00670488).
©2014 American Association for Cancer Research.
Figures

References
-
- Engelman JA. Targeting PI3K signalling in cancer: opportunities, challenges and limitations. Nat Rev Cancer. 2009;9:550–62. - PubMed
-
- Yap TA, Garrett MD, Walton MI, Raynaud F, de Bono JS, Workman P. Targeting the PI3K-AKT-mTOR pathway: progress, pitfalls, and promises. Curr Opin Pharmacol. 2008;8:393–412. - PubMed
-
- Jiao J, Wang S, Qiao R, Vivanco I, Watson PA, Sawyers CL, Wu H. Murine cell lines derived from Pten null prostate cancer show the critical role of PTEN in hormone refractory prostate cancer development. Cancer Res. 2007;67:6083–91. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

