Alzheimer presenilin-1 mutations dramatically reduce trimming of long amyloid β-peptides (Aβ) by γ-secretase to increase 42-to-40-residue Aβ
- PMID: 25239621
- PMCID: PMC4223309
- DOI: 10.1074/jbc.M114.581165
Alzheimer presenilin-1 mutations dramatically reduce trimming of long amyloid β-peptides (Aβ) by γ-secretase to increase 42-to-40-residue Aβ
Abstract
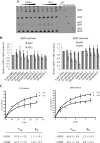
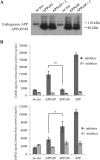
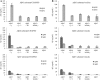
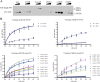
The presenilin-containing γ-secretase complex produces the amyloid β-peptide (Aβ) through intramembrane proteolysis, and >100 presenilin mutations are associated with familial early-onset Alzheimer disease (AD). The question of whether these mutations result in AD through a gain or a loss of function remains highly controversial. Mutations in presenilins increase ratios of 42- to 40-residue Aβ critical to pathogenesis, but other Aβs of 38-49 residues are also formed by γ-secretase. Evidence in cells suggests the protease first cleaves substrate within the transmembrane domain at the ϵ site to form 48- or 49-residue Aβ. Subsequent cleavage almost every three residues from the C terminus is thought to occur along two pathways toward shorter secreted forms of Aβ: Aβ49 → Aβ46 → Aβ43 → Aβ40 and Aβ48 → Aβ45 → Aβ42 → Aβ38. Here we show that the addition of synthetic long Aβ peptides (Aβ45-49) directly into purified preparations of γ-secretase leads to the formation of Aβ40 and Aβ42 whether the protease complex is detergent-solubilized or reconstituted into lipid vesicles, and the ratios of products Aβ42 to Aβ40 follow a pattern consistent with the dual-pathway hypothesis. Kinetic analysis of five different AD-causing mutations in presenilin-1 revealed that all result in drastic reduction of normal carboxypeptidase function. Altered trimming of long Aβ peptides to Aβ40 and Aβ42 by mutant proteases occurs at multiple levels, independent of the effects on initial endoproteolysis at the ϵ site, all conspiring to increase the critical Aβ42/Aβ40 ratio implicated in AD pathogenesis. Taken together, these results suggest that specific reduction of carboxypeptidase function of γ-secretase leads to the gain of toxic Aβ42/Aβ40.
Keywords: Amyloid-β (AB); Carboxypeptidase; Enzyme Kinetics; Enzyme Mechanism; Intramembrane Proteolysis.
© 2014 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures

Similar articles
-
Familial Alzheimer's disease mutations in amyloid protein precursor alter proteolysis by γ-secretase to increase amyloid β-peptides of ≥45 residues.J Biol Chem. 2021 Jan-Jun;296:100281. doi: 10.1016/j.jbc.2021.100281. Epub 2021 Jan 12. J Biol Chem. 2021. PMID: 33450230 Free PMC article.
-
Dissociation between the processivity and total activity of γ-secretase: implications for the mechanism of Alzheimer's disease-causing presenilin mutations.Biochemistry. 2011 Oct 25;50(42):9023-35. doi: 10.1021/bi2007146. Epub 2011 Sep 30. Biochemistry. 2011. PMID: 21919498 Free PMC article.
-
Presenilin-1 Familial Alzheimer Mutations Impair γ-Secretase Cleavage of APP Through Stabilized Enzyme-Substrate Complex Formation.Biomolecules. 2025 Jul 1;15(7):955. doi: 10.3390/biom15070955. Biomolecules. 2025. PMID: 40723827 Free PMC article.
-
Presenilin, γ-Secretase, and the Search for Pathogenic Triggers of Alzheimer's Disease.Biochemistry. 2025 Apr 15;64(8):1662-1672. doi: 10.1021/acs.biochem.4c00830. Epub 2025 Feb 25. Biochemistry. 2025. PMID: 39996369 Review.
-
Processive proteolysis by γ-secretase and the mechanism of Alzheimer's disease.Biol Chem. 2012 Sep;393(9):899-905. doi: 10.1515/hsz-2012-0140. Biol Chem. 2012. PMID: 22944690 Review.
Cited by
-
Interrelationship between Changes in the Amyloid β 42/40 Ratio and Presenilin 1 Conformation.Mol Med. 2016 Sep;22:329-337. doi: 10.2119/molmed.2016.00127. Epub 2016 Jul 5. Mol Med. 2016. PMID: 27391800 Free PMC article.
-
A thermodynamic investigation of amyloid precursor protein processing by human γ-secretase.Commun Biol. 2022 Aug 18;5(1):837. doi: 10.1038/s42003-022-03818-7. Commun Biol. 2022. PMID: 35982231 Free PMC article.
-
Extracellular interface between APP and Nicastrin regulates Aβ length and response to γ-secretase modulators.EMBO J. 2019 Jun 17;38(12):e101494. doi: 10.15252/embj.2019101494. Epub 2019 May 20. EMBO J. 2019. PMID: 31109937 Free PMC article.
-
Analysis of 138 pathogenic mutations in presenilin-1 on the in vitro production of Aβ42 and Aβ40 peptides by γ-secretase.Proc Natl Acad Sci U S A. 2017 Jan 24;114(4):E476-E485. doi: 10.1073/pnas.1618657114. Epub 2016 Dec 5. Proc Natl Acad Sci U S A. 2017. PMID: 27930341 Free PMC article.
-
Effect of Presenilin Mutations on APP Cleavage; Insights into the Pathogenesis of FAD.Front Aging Neurosci. 2016 Mar 11;8:51. doi: 10.3389/fnagi.2016.00051. eCollection 2016. Front Aging Neurosci. 2016. PMID: 27014058 Free PMC article.
References
-
- Goedert M., Spillantini M. G. (2006) A century of Alzheimer's disease. Science 314, 777–781 - PubMed
-
- Tanzi R. E., Bertram L. (2005) Twenty years of the Alzheimer's disease amyloid hypothesis: a genetic perspective. Cell 120, 545–555 - PubMed
-
- Wolfe M. S., Xia W., Ostaszewski B. L., Diehl T. S., Kimberly W. T., Selkoe D. J. (1999) Two transmembrane aspartates in presenilin-1 required for presenilin endoproteolysis and γ-secretase activity. Nature 398, 513–517 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

