Amphipathic tail-anchoring peptide is a promising therapeutic agent for prostate cancer treatment
- PMID: 25245280
- PMCID: PMC4202157
- DOI: 10.18632/oncotarget.2301
Amphipathic tail-anchoring peptide is a promising therapeutic agent for prostate cancer treatment
Abstract
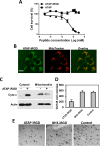
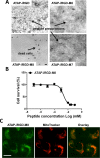
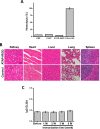
Amphipathic tail-anchoring peptide (ATAP) derived from the human anti-apoptotic protein Bfl-1 is a potent inducer of apoptosis by targeting mitochondria permeability transition. By linking ATAP to an internalizing RGD peptide (iRGD), selective targeting for ATAP to tumor cell was achieved. Confocal fluorescence microscopy showed that ATAP-iRGD could penetrate into cancer cells and distribute along the mitochondria network. ATAP-iRGD triggered mitochondria-dependent cell death through release of cytochrome c. In an effort to promote ATAP-iRGD physiochemical properties to approach clinic application, amino acid substitution and chemical modification were made with ATAP-iRGD to improve its bioactivity. One of these modified peptides, ATAP-iRGD-M8, was with improved stability and aqueous solubility without compromising in vitro cytotoxicity in cultured cancer cells. In vivo xenograft studies with multiple prostate cancer cell lines showed that intravenous administration of ATAP-iRGD-M8 suppressed tumor growth. Toxicological studies revealed that repetitive intravenous administration of ATAP-iRGD-M8 did not produce significant toxicity in the SV129 mice. Our data suggest that ATAP-iRGD-M8 is a promising agent with high selectivity and limited systemic toxicity for prostate cancer treatment.
Conflict of interest statement
JM has an equity interest in TRIM–edicine, which develops ATAP as potential therapeutic agent for cancer treatment. Patents on the use of ATAP are held by the Rutgers University—Robert Wood Johnson Medical School.
Figures


Similar articles
-
Amphipathic tail-anchoring peptide and Bcl-2 homology domain-3 (BH3) peptides from Bcl-2 family proteins induce apoptosis through different mechanisms.J Biol Chem. 2011 Mar 18;286(11):9038-48. doi: 10.1074/jbc.M110.198457. Epub 2010 Dec 28. J Biol Chem. 2011. PMID: 21189256 Free PMC article.
-
The tail-anchoring domain of Bfl1 and HCCS1 targets mitochondrial membrane permeability to induce apoptosis.J Cell Sci. 2007 Aug 15;120(Pt 16):2912-23. doi: 10.1242/jcs.006197. Epub 2007 Jul 31. J Cell Sci. 2007. PMID: 17666431
-
iRGD-targeted delivery of a pro-apoptotic peptide activated by cathepsin B inhibits tumor growth and metastasis in mice.Tumour Biol. 2016 Aug;37(8):10643-52. doi: 10.1007/s13277-016-4961-x. Epub 2016 Feb 11. Tumour Biol. 2016. PMID: 26867766
-
Cancer therapy with iRGD as a tumor-penetrating peptide.Bull Cancer. 2023 Dec;110(12):1288-1300. doi: 10.1016/j.bulcan.2023.08.009. Epub 2023 Oct 8. Bull Cancer. 2023. PMID: 37813754 Review.
-
iRGD as a tumor‑penetrating peptide for cancer therapy (Review).Mol Med Rep. 2017 May;15(5):2925-2930. doi: 10.3892/mmr.2017.6419. Epub 2017 Mar 30. Mol Med Rep. 2017. PMID: 28358432 Review.
Cited by
-
MG53 suppresses tumor progression and stress granule formation by modulating G3BP2 activity in non-small cell lung cancer.Mol Cancer. 2021 Sep 14;20(1):118. doi: 10.1186/s12943-021-01418-3. Mol Cancer. 2021. PMID: 34521423 Free PMC article.
-
Coadministration of kla peptide with HPRP-A1 to enhance anticancer activity.PLoS One. 2019 Nov 8;14(11):e0223738. doi: 10.1371/journal.pone.0223738. eCollection 2019. PLoS One. 2019. PMID: 31703065 Free PMC article.
-
Core-shell nanoparticle-based peptide therapeutics and combined hyperthermia for enhanced cancer cell apoptosis.ACS Nano. 2014 Sep 23;8(9):9379-87. doi: 10.1021/nn503431x. Epub 2014 Aug 21. ACS Nano. 2014. PMID: 25133971 Free PMC article.
-
Targeting Tumors Using Peptides.Molecules. 2020 Feb 13;25(4):808. doi: 10.3390/molecules25040808. Molecules. 2020. PMID: 32069856 Free PMC article. Review.
-
Peptides as drug delivery vehicles across biological barriers.J Pharm Investig. 2018 Jan;48(1):89-111. doi: 10.1007/s40005-017-0374-0. Epub 2017 Dec 12. J Pharm Investig. 2018. PMID: 29963321 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

