Platelet mechanosensing of substrate stiffness during clot formation mediates adhesion, spreading, and activation
- PMID: 25246564
- PMCID: PMC4210024
- DOI: 10.1073/pnas.1322917111
Platelet mechanosensing of substrate stiffness during clot formation mediates adhesion, spreading, and activation
Abstract
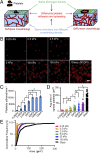
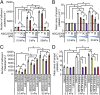
As platelets aggregate and activate at the site of vascular injury to stem bleeding, they are subjected to a myriad of biochemical and biophysical signals and cues. As clot formation ensues, platelets interact with polymerizing fibrin scaffolds, exposing platelets to a large range of mechanical microenvironments. Here, we show for the first time (to our knowledge) that platelets, which are anucleate cellular fragments, sense microenvironmental mechanical properties, such as substrate stiffness, and transduce those cues into differential biological signals. Specifically, as platelets mechanosense the stiffness of the underlying fibrin/fibrinogen substrate, increasing substrate stiffness leads to increased platelet adhesion and spreading. Importantly, adhesion on stiffer substrates also leads to higher levels of platelet activation, as measured by integrin αIIbβ3 activation, α-granule secretion, and procoagulant activity. Mechanistically, we determined that Rac1 and actomyosin activity mediate substrate stiffness-dependent platelet adhesion, spreading, and activation to different degrees. This capability of platelets to mechanosense microenvironmental cues in a growing thrombus or hemostatic plug and then mechanotransduce those cues into differential levels of platelet adhesion, spreading, and activation provides biophysical insight into the underlying mechanisms of platelet aggregation and platelet activation heterogeneity during thrombus formation.
Keywords: biomaterials; cell mechanics; mechanotransduction; platelet cytoskeleton.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Jackson SP. Arterial thrombosis—insidious, unpredictable and deadly. Nat Med. 2011;17(11):1423–1436. - PubMed
-
- Ruggeri ZM. Platelets in atherothrombosis. Nat Med. 2002;8(11):1227–1234. - PubMed
-
- Shattil SJ, Kashiwagi H, Pampori N. Integrin signaling: The platelet paradigm. Blood. 1998;91(8):2645–2657. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

