Vaginal self-sampling is a cost-effective way to increase participation in a cervical cancer screening programme: a randomised trial
- PMID: 25247320
- PMCID: PMC4260034
- DOI: 10.1038/bjc.2014.510
Vaginal self-sampling is a cost-effective way to increase participation in a cervical cancer screening programme: a randomised trial
Abstract
Background: Cervical cancer screening coverage remains insufficient in most countries. Our objective was to assess whether in-home vaginal self-sampling with a dry swab for high-risk human papillomavirus (HR-HPV) testing is effective and cost-effective in increasing participation in cervical cancer screening.
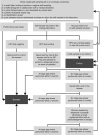
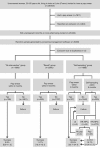
Methods: In March 2012, 6000 unscreened women aged 30-65 years, living in a French region covered by a screening programme, who had not responded to an initial invitation to have a Pap smear were equally randomised to three groups: 'no intervention'; 'recall', women received a letter to have a Pap smear; and 'self-sampling', women received a self-sampling kit to return to a centralised virology laboratory for PCR-based HPV testing.
Results: Participation was higher in the 'self-sampling' than in the 'no intervention' group (22.5% vs 9.9%, P<0.0001; OR 2.64) and 'recall' group (11.7%, P<0.0001; OR 2.20). In the 'self-sampling' group, 320 used the self-sampling kit; for 44 of these women with positive HR-HPV test results, 40 had the recommended triage Pap smear. The ICER per extra screened woman was 77.8[euro ] and 63.2[euro ] for the 'recall' and 'self-sampling' groups, respectively, relative to the 'no intervention' group.
Conclusions: Offering an in-home, return-mail kit for vaginal self-sampling with a dry swab is more effective and cost-effective than a recall letter in increasing participation in cervical cancer screening.
Figures
References
-
- Arbyn M, Raifu AO, Weiderpass E, Bray F, Anttila A. Trends of cervical cancer mortality in the member states of the European Union. Eur J Cancer. 2009;45:2640–2648. - PubMed
-
- Arbyn M, Ronco G, Anttila A, CJLM Meijer, Poljak M, Ogilvie G, Koliopoulos G, Naucler P, Sankaranarayanan R, Peto J. Evidence regarding human papillomavirus testing in secondary prevention of cervical cancer. Vaccine. 2012;30 (Suppl 5:F88–F99. - PubMed
-
- Arbyn M, Verdoodt F, Snijders PJF, Verhoef VMJ, Suonio E, Dillner L, Minozzi S, Bellisario C, Banzi R, Zhao F-H, Hillemanns P, Anttila A. Accuracy of human papillomavirus testing on self-collected versus clinician-collected samples: a meta-analysis. Lancet Oncol. 2014;15:172–183. - PubMed
-
- Van Baars R, Bosgraaf RP, ter Harmsel BWA, Melchers WJG, Quint WGV, Bekkers RLM. Dry storage and transport of a cervicovaginal self-sample by use of the Evalyn Brush, providing reliable human papillomavirus detection combined with comfort for women. J Clin Microbiol. 2012;50:3937–3943. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

