Matrix metalloproteinase-19 promotes metastatic behavior in vitro and is associated with increased mortality in non-small cell lung cancer
- PMID: 25250855
- PMCID: PMC4299607
- DOI: 10.1164/rccm.201310-1903OC
Matrix metalloproteinase-19 promotes metastatic behavior in vitro and is associated with increased mortality in non-small cell lung cancer
Abstract
Rationale: Lung cancer is the leading cause of cancer death in both men and women in the United States and worldwide. Matrix metalloproteinases (MMPs) have been implicated in the development and progression of lung cancer, but their role in the molecular pathogenesis of lung cancer remains unclear. We have found that MMP19, a relatively novel member of the MMP family, is overexpressed in lung tumors when compared with control subjects.
Objectives: To test the hypothesis that MMP19 plays a significant role in the development and progression of non-small cell lung cancer (NSCLC).
Methods: We have analyzed lung cancer gene expression data, immunostained lung tumors for MMP19, and performed in vitro assays to test the effects of MMP19 in NSCLC cells.
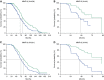
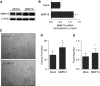
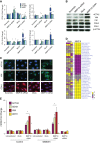
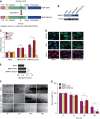
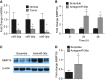
Measurements and main results: We found that MMP19 gene and protein expression is increased in lung cancer tumors compared with adjacent and histologically normal lung tissues. In three independent datasets, increased MMP19 gene expression conferred a poorer prognosis in NSCLC. In vitro, we found that overexpression of MMP19 promotes epithelial-mesenchymal transition, migration, and invasiveness in multiple NSCLC cell lines. Overexpression of MMP19 with a mutation at the catalytic site did not impair epithelial-mesenchymal transition or expression of prometastasis genes. We also found that miR-30 isoforms, a microRNA family predicted to target MMP19, is markedly down-regulated in human lung cancer and regulates MMP19 expression.
Conclusions: Taken together, these findings suggest that MMP19 is associated with the development and progression of NSCLC and may be a potential biomarker of disease severity and outcome.
Keywords: MMP19; epithelial–mesenchymal transition; metastasis; miR-30; non–small cell lung cancer.
Figures


Similar articles
-
MCRS1 overexpression, which is specifically inhibited by miR-129*, promotes the epithelial-mesenchymal transition and metastasis in non-small cell lung cancer.Mol Cancer. 2014 Nov 6;13:245. doi: 10.1186/1476-4598-13-245. Mol Cancer. 2014. PMID: 25373388 Free PMC article.
-
MicroRNA-16 inhibits the proliferation, migration and invasion of non-small cell lung carcinoma cells by down-regulating matrix metalloproteinase-19 expression.Eur Rev Med Pharmacol Sci. 2019 Jun;23(12):5260-5269. doi: 10.26355/eurrev_201906_18192. Eur Rev Med Pharmacol Sci. 2019. PMID: 31298377
-
UBE2C, Directly Targeted by miR-548e-5p, Increases the Cellular Growth and Invasive Abilities of Cancer Cells Interacting with the EMT Marker Protein Zinc Finger E-box Binding Homeobox 1/2 in NSCLC.Theranostics. 2019 Mar 17;9(7):2036-2055. doi: 10.7150/thno.32738. eCollection 2019. Theranostics. 2019. Retraction in: Theranostics. 2020 Jul 25;10(21):9619. doi: 10.7150/thno.50254. PMID: 31037155 Free PMC article. Retracted.
-
Long non-coding RNA PRNCR1 modulates non-small cell lung cancer cell proliferation, apoptosis, migration, invasion, and EMT through PRNCR1/miR-126-5p/MTDH axis.Biosci Rep. 2020 Jul 31;40(7):BSR20193153. doi: 10.1042/BSR20193153. Biosci Rep. 2020. PMID: 31912882 Free PMC article.
-
Cellular Senescence in Non-Small Cell Lung Cancer.Front Biosci (Landmark Ed). 2023 Dec 28;28(12):357. doi: 10.31083/j.fbl2812357. Front Biosci (Landmark Ed). 2023. PMID: 38179738 Review.
Cited by
-
The multifaceted roles of matrix metalloproteinases in lung cancer.Front Oncol. 2023 Sep 12;13:1195426. doi: 10.3389/fonc.2023.1195426. eCollection 2023. Front Oncol. 2023. PMID: 37766868 Free PMC article. Review.
-
IL-17 induces NSCLC cell migration and invasion by elevating MMP19 gene transcription and expression through the interaction of p300-dependent STAT3-K631 acetylation and its Y705-phosphorylation.Oncol Res. 2024 Mar 20;32(4):625-641. doi: 10.32604/or.2023.031053. eCollection 2024. Oncol Res. 2024. PMID: 38560562 Free PMC article.
-
HSPB8 Facilitates the Oncogenesis and Advancement of Bladder Cancer via Activation of HSP27.J Cancer. 2024 Jan 1;15(3):645-658. doi: 10.7150/jca.89994. eCollection 2024. J Cancer. 2024. PMID: 38213722 Free PMC article.
-
A critical ETV4/Twist1/Vimentin axis in Ha-RAS-induced aggressive breast cancer.Cancer Gene Ther. 2022 Nov;29(11):1590-1599. doi: 10.1038/s41417-022-00471-4. Epub 2022 Apr 27. Cancer Gene Ther. 2022. PMID: 35477769
-
miR-30 Family Reduction Maintains Self-Renewal and Promotes Tumorigenesis in NSCLC-Initiating Cells by Targeting Oncogene TM4SF1.Mol Ther. 2018 Dec 5;26(12):2751-2765. doi: 10.1016/j.ymthe.2018.09.006. Epub 2018 Sep 13. Mol Ther. 2018. PMID: 30301667 Free PMC article.
References
-
- United States Cancer Statistics1999–2009 Incidence and Mortality Web-based Report. Department of Health and Human Services, Centers for Disease Control and Prevention, and National Cancer Institute, 2013[accessed 2013 Mar 6]. Available from: http://www.cdc.gov/cancer/lung/statistics/index.htm
-
- Surveillance, Epidemiology, and End Results ProgramTurning cancer data into discovery. National Cancer Institute, 2013[accessed 2014 Apr 3]. Available from: http://seer.cancer.gov
-
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. - PubMed
-
- Khokha R, Murthy A, Weiss A. Metalloproteinases and their natural inhibitors in inflammation and immunity. Nat Rev Immunol. 2013;13:649–665. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

