Shc depletion stimulates brown fat activity in vivo and in vitro
- PMID: 25257068
- PMCID: PMC4244234
- DOI: 10.1111/acel.12267
Shc depletion stimulates brown fat activity in vivo and in vitro
Abstract
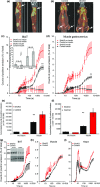
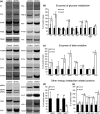
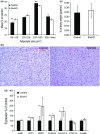
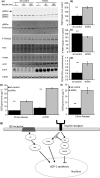
Adipose tissue is an important metabolic organ that integrates a wide array of homeostatic processes and is crucial for whole-body insulin sensitivity and energy metabolism. Brown adipose tissue (BAT) is a key thermogenic tissue with a well-established role in energy expenditure. BAT dissipates energy and protects against both hypothermia and obesity. Thus, BAT stimulation therapy is a rational strategy for the looming pandemic of obesity, whose consequences and comorbidities have a huge impact on the aged. Shc-deficient mice (ShcKO) were previously shown to be lean, insulin sensitive, and resistant to high-fat diet and obesity. We investigated the contribution of BAT to this phenotype. Insulin-dependent BAT glucose uptake was higher in ShcKO mice. Primary ShcKO BAT cells exhibited increased mitochondrial respiration; increased expression of several mitochondrial and lipid-oxidative enzymes was observed in ShcKO BAT. Levels of brown fat-specific markers of differentiation, UCP1, PRDM16, ELOVL3, and Cox8b, were higher in ShcKO BAT. In vitro, Shc knockdown in BAT cell line increased insulin sensitivity and metabolic activity. In vivo, pharmacological stimulation of ShcKO BAT resulted in higher energy expenditure. Conversely, pharmacological inhibition of BAT abolished the improved metabolic parameters, that is the increased insulin sensitivity and glucose tolerance of ShcKO mice. Similarly, in vitro Shc knockdown in BAT cell lines increased their expression of UCP1 and metabolic activity. These data suggest increased BAT activity significantly contributes to the improved metabolic phenotype of ShcKO mice.
Keywords: Shc; brown adipose; brown adipose tissue; energy expenditure; healthy aging; insulin.
© 2014 The Authors. Aging Cell published by the Anatomical Society and John Wiley & Sons Ltd.
Figures


References
-
- Ahfeldt T, Schinzel RT, Lee YK, Hendrickson D, Kaplan A, Lum DH, Camahort R, Xia F, Shay J, Rhee EP, Clish CB, Deo RC, Shen T, Lau FH, Cowley A, Mowrer G, Al-Siddiqi H, Nahrendorf M, Musunuru K, Gerszten RE, Rinn JL, Cowan CA. Programming human pluripotent stem cells into white and brown adipocytes. Nat. Cell Biol. 2012;14:209–219. - PMC - PubMed
-
- Boney CM, Sekimoto H, Gruppuso PA, Frackelton AR., Jr Src family tyrosine kinases participate in insulin-like growth factor I mitogenic signaling in 3T3-L1 cells. Cell Growth Differ. 2001;12:379–386. - PubMed
-
- Camici GG, Schiavoni M, Francia P, Bachschmid M, Martin-Padura I, Hersberger M, Tanner FC, Pelicci P, Volpe M, Anversa P, Luscher TF, Cosentino F. Genetic deletion of p66(Shc) adaptor protein prevents hyperglycemia-induced endothelial dysfunction and oxidative stress. Proc. Natl Acad. Sci. USA. 2007;104:5217–5222. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

