An acute rise in intraluminal pressure shifts the mediator of flow-mediated dilation from nitric oxide to hydrogen peroxide in human arterioles
- PMID: 25260615
- PMCID: PMC4255007
- DOI: 10.1152/ajpheart.00557.2014
An acute rise in intraluminal pressure shifts the mediator of flow-mediated dilation from nitric oxide to hydrogen peroxide in human arterioles
Abstract
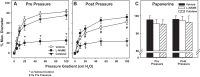
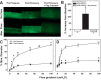
Endothelial nitric oxide (NO) is the primary mediator of flow-mediated dilation (FMD) in human adipose microvessels. Impaired NO-mediated vasodilation occurs after acute and chronic hypertension, possibly due to excess generation of reactive oxygen species (ROS). The direct role of pressure elevation in this impairment of human arteriolar dilation is not known. We tested the hypothesis that elevation in pressure is sufficient to impair FMD. Arterioles were isolated from human adipose tissue and cannulated, and vasodilation to graded flow gradients was measured before and after exposure to increased intraluminal pressure (IILP; 150 mmHg, 30 min). The mediator of FMD was determined using pharmacological agents to reduce NO [N(G)-nitro-l-arginine methyl ester (l-NAME), 2-(4-carboxyphenyl)-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide (c-PTIO)], or H2O2 [polyethylene glycol (PEG)-catalase], and mitochondrial (mt) ROS was quantified using fluorescence microscopy. Exposure to IILP decreased overall FMD (max %dilation: 82.7 ± 4.9 vs. 62 ± 5.6; P < 0.05). This dilation was abolished by treatment with l-NAME prepressure and PEG-catalase after IILP (max %dilation: l-NAME: 23.8 ± 6.1 vs. 74.8 ± 8.6; PEG-catalase: 71.8 ± 5.9 vs. 24.6 ± 10.6). To examine if this change was mediated by mtROS, FMD responses were measured in the presence of the complex I inhibitor rotenone or the mitochondrial antioxidant mitoTempol. Before IILP, FMD was unaffected by either compound; however, both inhibited dilation after IILP. The fluorescence intensity of mitochondria peroxy yellow 1 (MitoPY1), a mitochondria-specific fluorescent probe for H2O2, increased during flow after IILP (%change from static: 12.3 ± 14.5 vs. 127.9 ± 57.7). These results demonstrate a novel compensatory dilator mechanism in humans that is triggered by IILP, inducing a change in the mediator of FMD from NO to mitochondria-derived H2O2.
Keywords: flow-mediated dilation; mitochondria; reactive oxygen species; vascular endothelium.
Copyright © 2014 the American Physiological Society.
Figures


References
-
- Channon K. Tetrahydrobiopterin: regulator of endothelial nitric oxide synthase in vascular disease. Trends Cardiovasc Med 14: 323–327, 2004. - PubMed
-
- Chlopicki S, Kozlovski VI, Lorkowska B, Drelicharz L, Gebska A. Compensation of endothelium-dependent responses in coronary circulation of eNOS-deficient mice. J Cardiovasc Pharmacol 46: 115–123, 2005. - PubMed
-
- Cooke M, John P, Dzau M, Victor J. Nitric oxide synthase: role in the genesis of vascular disease. Annu Rev Med 48: 489–509, 1997. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

