The convergence of fracture repair and stem cells: interplay of genes, aging, environmental factors and disease
- PMID: 25264148
- PMCID: PMC4455538
- DOI: 10.1002/jbmr.2373
The convergence of fracture repair and stem cells: interplay of genes, aging, environmental factors and disease
Abstract
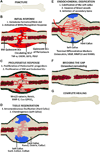
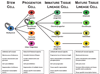
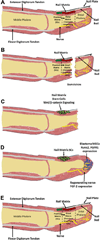
The complexity of fracture repair makes it an ideal process for studying the interplay between the molecular, cellular, tissue, and organ level events involved in tissue regeneration. Additionally, as fracture repair recapitulates many of the processes that occur during embryonic development, investigations of fracture repair provide insights regarding skeletal embryogenesis. Specifically, inflammation, signaling, gene expression, cellular proliferation and differentiation, osteogenesis, chondrogenesis, angiogenesis, and remodeling represent the complex array of interdependent biological events that occur during fracture repair. Here we review studies of bone regeneration in genetically modified mouse models, during aging, following environmental exposure, and in the setting of disease that provide insights regarding the role of multipotent cells and their regulation during fracture repair. Complementary animal models and ongoing scientific discoveries define an increasing number of molecular and cellular targets to reduce the morbidity and complications associated with fracture repair. Last, some new and exciting areas of stem cell research such as the contribution of mitochondria function, limb regeneration signaling, and microRNA (miRNA) posttranscriptional regulation are all likely to further contribute to our understanding of fracture repair as an active branch of regenerative medicine.
Keywords: AGING; ANIMAL MODELS; CELL/TISSUE SIGNALING; CELLS OF BONE; GENETICALLY ALTERED MICE; INJURY/FRACTURE HEALING; ORTHOPAEDICS; PARACRINE PATHWAYS; STEM AND PROGENITOR CELLS.
© 2014 American Society for Bone and Mineral Research.
Conflict of interest statement
RJO provides consulting services for GlaxoSmithKline. MH has no conflicts of interest.
Figures
Similar articles
-
Reduced COX-2 expression in aged mice is associated with impaired fracture healing.J Bone Miner Res. 2009 Feb;24(2):251-64. doi: 10.1359/jbmr.081002. J Bone Miner Res. 2009. PMID: 18847332 Free PMC article.
-
Age-related inflammation triggers skeletal stem/progenitor cell dysfunction.Proc Natl Acad Sci U S A. 2019 Apr 2;116(14):6995-7004. doi: 10.1073/pnas.1810692116. Epub 2019 Mar 20. Proc Natl Acad Sci U S A. 2019. PMID: 30894483 Free PMC article.
-
Prospects for osteoprogenitor stem cells in fracture repair and osteoporosis.Curr Opin Organ Transplant. 2010 Feb;15(1):73-8. doi: 10.1097/MOT.0b013e328333d52c. Curr Opin Organ Transplant. 2010. PMID: 19935065 Free PMC article. Review.
-
The spatiotemporal role of COX-2 in osteogenic and chondrogenic differentiation of periosteum-derived mesenchymal progenitors in fracture repair.PLoS One. 2014 Jul 2;9(7):e100079. doi: 10.1371/journal.pone.0100079. eCollection 2014. PLoS One. 2014. PMID: 24988184 Free PMC article.
-
Fracture healing: mechanisms and interventions.Nat Rev Rheumatol. 2015 Jan;11(1):45-54. doi: 10.1038/nrrheum.2014.164. Epub 2014 Sep 30. Nat Rev Rheumatol. 2015. PMID: 25266456 Free PMC article. Review.
Cited by
-
Regional variations of jaw bone characteristics in an ovariectomized rat model.J Mech Behav Biomed Mater. 2020 Oct;110:103952. doi: 10.1016/j.jmbbm.2020.103952. Epub 2020 Jun 29. J Mech Behav Biomed Mater. 2020. PMID: 32957244 Free PMC article.
-
Differential fracture response to traumatic brain injury suggests dominance of neuroinflammatory response in polytrauma.Sci Rep. 2019 Aug 21;9(1):12199. doi: 10.1038/s41598-019-48126-z. Sci Rep. 2019. PMID: 31434912 Free PMC article.
-
Emergent properties of neural repair: elemental biology to therapeutic concepts.Ann Neurol. 2016 Jun;79(6):895-906. doi: 10.1002/ana.24653. Epub 2016 Apr 21. Ann Neurol. 2016. PMID: 27043816 Free PMC article. Review.
-
Different Protocols of Combined Application of Photobiomodulation In Vitro and In Vivo Plus Adipose-Derived Stem Cells Improve the Healing of Bones in Critical Size Defects in Rat Models.J Lasers Med Sci. 2022 Mar 5;13:e10. doi: 10.34172/jlms.2022.10. eCollection 2022. J Lasers Med Sci. 2022. PMID: 35996492 Free PMC article.
-
PTH 1-34 Ameliorates the Osteopenia and Delayed Healing of Stabilized Tibia Fracture in Mice with Achondroplasia Resulting from Gain-Of-Function Mutation of FGFR3.Int J Biol Sci. 2017 Sep 21;13(10):1254-1265. doi: 10.7150/ijbs.21258. eCollection 2017. Int J Biol Sci. 2017. PMID: 29104492 Free PMC article.
References
-
- Mabry TM, Prpa B, Haidukewych GJ, Harmsen WS, Berry DJ. Long-term results of total hip arthroplasty for femoral neck fracture nonunion. J Bone Joint Surg Am. 2004;86-A(10):2263–2267. - PubMed
-
- Frolke JP, Patka P. Definition and classification of fracture nonunions. Injury. 2007;38(Suppl 2):S19–S22. - PubMed
-
- Ryzewicz M, Morgan SJ, Linford E, Thwing JI, de Resende GV, Smith WR. Central bone grafting for nonunion of fractures of the tibia: a retrospective series. J Bone Joint Surg Br. 2009;91(4):522–529. - PubMed
-
- Lin J. Effectiveness of completely round nails with both-ends-threaded locking screws for tibial shaft fractures. J Trauma. 2006;61(4):893–899. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

