Memory maintenance in synapses with calcium-based plasticity in the presence of background activity
- PMID: 25275319
- PMCID: PMC4183374
- DOI: 10.1371/journal.pcbi.1003834
Memory maintenance in synapses with calcium-based plasticity in the presence of background activity
Abstract
Most models of learning and memory assume that memories are maintained in neuronal circuits by persistent synaptic modifications induced by specific patterns of pre- and postsynaptic activity. For this scenario to be viable, synaptic modifications must survive the ubiquitous ongoing activity present in neural circuits in vivo. In this paper, we investigate the time scales of memory maintenance in a calcium-based synaptic plasticity model that has been shown recently to be able to fit different experimental data-sets from hippocampal and neocortical preparations. We find that in the presence of background activity on the order of 1 Hz parameters that fit pyramidal layer 5 neocortical data lead to a very fast decay of synaptic efficacy, with time scales of minutes. We then identify two ways in which this memory time scale can be extended: (i) the extracellular calcium concentration in the experiments used to fit the model are larger than estimated concentrations in vivo. Lowering extracellular calcium concentration to in vivo levels leads to an increase in memory time scales of several orders of magnitude; (ii) adding a bistability mechanism so that each synapse has two stable states at sufficiently low background activity leads to a further boost in memory time scale, since memory decay is no longer described by an exponential decay from an initial state, but by an escape from a potential well. We argue that both features are expected to be present in synapses in vivo. These results are obtained first in a single synapse connecting two independent Poisson neurons, and then in simulations of a large network of excitatory and inhibitory integrate-and-fire neurons. Our results emphasise the need for studying plasticity at physiological extracellular calcium concentration, and highlight the role of synaptic bi- or multistability in the stability of learned synaptic structures.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
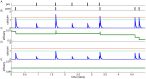
 and
and  ; see Table 1). Whenever the calcium trace crosses the depression (cyan) or potentiation thresholds (orange), changes in the synaptic efficacy (green) are induced. (D,E) Same as in B,C but with calcium amplitudes corresponding to the in vivo case (
; see Table 1). Whenever the calcium trace crosses the depression (cyan) or potentiation thresholds (orange), changes in the synaptic efficacy (green) are induced. (D,E) Same as in B,C but with calcium amplitudes corresponding to the in vivo case ( and
and  ). The small calcium transients do not cross the depression/potentiation thresholds and no efficacy changes are observed. Note that the flat potential for
). The small calcium transients do not cross the depression/potentiation thresholds and no efficacy changes are observed. Note that the flat potential for  is used here and that noise is set to zero for clarity,
is used here and that noise is set to zero for clarity,  .
.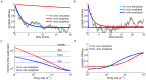
 synapses). Blue and red lines show the mean dynamics as predicted by the Ornstein-Uhlenbeck theory. Grey lines show example traces of synaptic efficacy evolution in time. (C) Decay time constant as a function of the firing rate for in vitro and in vivo parameter sets. The blue and red lines show the calculated decay time constant,
synapses). Blue and red lines show the mean dynamics as predicted by the Ornstein-Uhlenbeck theory. Grey lines show example traces of synaptic efficacy evolution in time. (C) Decay time constant as a function of the firing rate for in vitro and in vivo parameter sets. The blue and red lines show the calculated decay time constant,  , from the OU theory. The points show exponential decay times obtained by fitting single exponential decay functions to the mean synaptic dynamics as shown in A and B illustrating that the OU theory correctly describes the full model behaviour. The cyan and orange dotted lines illustrate the derived power law behaviour,
, from the OU theory. The points show exponential decay times obtained by fitting single exponential decay functions to the mean synaptic dynamics as shown in A and B illustrating that the OU theory correctly describes the full model behaviour. The cyan and orange dotted lines illustrate the derived power law behaviour,  , between memory time scales and low firing rates (see text). The power reflects the number of spikes required to cross the plasticity thresholds, that is,
, between memory time scales and low firing rates (see text). The power reflects the number of spikes required to cross the plasticity thresholds, that is,  for in vitro (cyan dotted line) and
for in vitro (cyan dotted line) and  (orange dotted line) for in vivo case. (D) Asymptotic synaptic efficacy as a function of the firing rate for in vitro and in vivo parameter sets. The lines show the calculated asymptotic value,
(orange dotted line) for in vivo case. (D) Asymptotic synaptic efficacy as a function of the firing rate for in vitro and in vivo parameter sets. The lines show the calculated asymptotic value,  , from the truncated OU theory (
, from the truncated OU theory ( ) for in vitro (blue line) and in vivo (red line) cases. Note that at high frequencies
) for in vitro (blue line) and in vivo (red line) cases. Note that at high frequencies  saturates at a value equal to
saturates at a value equal to  , since both depression and potentiation terms are active in the high calcium region. The points show steady-state values obtained by fitting single exponential decay functions to the mean synaptic dynamics as shown in A and B (green: in vitro; light blue: in vivo).
, since both depression and potentiation terms are active in the high calcium region. The points show steady-state values obtained by fitting single exponential decay functions to the mean synaptic dynamics as shown in A and B (green: in vitro; light blue: in vivo).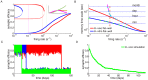
 /s for in vitro and
/s for in vitro and  /s for in vivo) and monostable at high firing rats. The effective potential of synaptic efficacy is shown for three firing rates (0.1/s - magenta line; 1/s - orange line; 2/s - green line) and the in vivo parameter set in the inset (firing rates indicated by vertical lines). (B) Decay time constant as a function of the firing rate for the in vitro and the in vivo parameter sets. For the in vivo parameter set below
/s for in vivo) and monostable at high firing rats. The effective potential of synaptic efficacy is shown for three firing rates (0.1/s - magenta line; 1/s - orange line; 2/s - green line) and the in vivo parameter set in the inset (firing rates indicated by vertical lines). (B) Decay time constant as a function of the firing rate for the in vitro and the in vivo parameter sets. For the in vivo parameter set below  /s, the bistability greatly extends memory time scale compared to a synapse with flat potential (red line) and can be predicted using Kramers escape rate (magenta line). The vertical dashed line illustrates the frequency at the in vivo bifurcation point. For the in vitro parameter set, the bistability has no influence on decay time constants for firing rates above 0.1/s. The points show exponential decay times obtained by fitting single exponential decay functions to the mean synaptic dynamics. (C) Individual synaptic efficacy traces for the in vivo parameter set at 1/s pre- and postsynaptic firing. The synapses remain in the upper potential well for a long time and stochastically cross the potential barrier to the low efficacy state. (D) Averaged synaptic efficacy trace of many synapses for the in-vivo parameter set at 1/s. The bistability extends the memory time scale from hours for a flat potential to days.
/s, the bistability greatly extends memory time scale compared to a synapse with flat potential (red line) and can be predicted using Kramers escape rate (magenta line). The vertical dashed line illustrates the frequency at the in vivo bifurcation point. For the in vitro parameter set, the bistability has no influence on decay time constants for firing rates above 0.1/s. The points show exponential decay times obtained by fitting single exponential decay functions to the mean synaptic dynamics. (C) Individual synaptic efficacy traces for the in vivo parameter set at 1/s pre- and postsynaptic firing. The synapses remain in the upper potential well for a long time and stochastically cross the potential barrier to the low efficacy state. (D) Averaged synaptic efficacy trace of many synapses for the in-vivo parameter set at 1/s. The bistability extends the memory time scale from hours for a flat potential to days.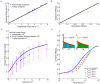
 , has a marked influence on the observed average synaptic efficacy. Depolarised/hyperpolarised reset potentials (e.g. −55/−70 mV, cyan/green dots) lead to an over/under-representation of short ISIs (left/right inset) compared to Poisson neurons (red line in insets). ISI histograms in inset are shown for LIF neurons firing at 1/s.
, has a marked influence on the observed average synaptic efficacy. Depolarised/hyperpolarised reset potentials (e.g. −55/−70 mV, cyan/green dots) lead to an over/under-representation of short ISIs (left/right inset) compared to Poisson neurons (red line in insets). ISI histograms in inset are shown for LIF neurons firing at 1/s.

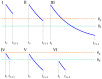
 (orange dashed line), and the depression thresholds,
(orange dashed line), and the depression thresholds,  (cyan dashed line).
(cyan dashed line).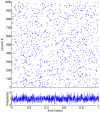
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

