Mechanism and function of mixed-mode oscillations in vibrissa motoneurons
- PMID: 25275462
- PMCID: PMC4183652
- DOI: 10.1371/journal.pone.0109205
Mechanism and function of mixed-mode oscillations in vibrissa motoneurons
Abstract
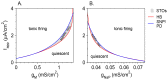
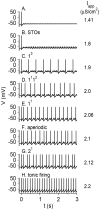
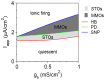
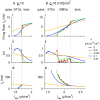
Vibrissa motoneurons in the facial nucleus innervate the intrinsic and extrinsic muscles that move the whiskers. Their intrinsic properties affect the way they process fast synaptic input from the vIRT and Bötzinger nuclei together with serotonergic neuromodulation. In response to constant current (I(app)) injection, vibrissa motoneurons may respond with mixed mode oscillations (MMOs), in which sub-threshold oscillations (STOs) are intermittently mixed with spikes. This study investigates the mechanisms involved in generating MMOs in vibrissa motoneurons and their function in motor control. It presents a conductance-based model that includes the M-type K+ conductance, g(M), the persistent Na+ conductance, g(NaP), and the cationic h conductance, g(h). For g(h) = 0 and moderate values of g(M) and g(NaP), the model neuron generates STOs, but not MMOs, in response to I(app) injection. STOs transform abruptly to tonic spiking as the current increases. In addition to STOs, MMOs are generated for g(h)>0 for larger values of I(app); the I(app) range in which MMOs appear increases linearly with g(h). In the MMOs regime, the firing rate increases with I(app) like a Devil's staircase. Stochastic noise disrupts the temporal structure of the MMOs, but for a moderate noise level, the coefficient of variation (CV) is much less than one and varies non-monotonically with I(app). Furthermore, the estimated time period between voltage peaks, based on Bernoulli process statistics, is much higher in the MMOs regime than in the tonic regime. These two phenomena do not appear when moderate noise generates MMOs without an intrinsic MMO mechanism. Therefore, and since STOs do not appear in spinal motoneurons, the analysis can be used to differentiate different MMOs mechanisms. MMO firing activity in vibrissa motoneurons suggests a scenario in which moderate periodic inputs from the vIRT and Bötzinger nuclei control whisking frequency, whereas serotonergic neuromodulation controls whisking amplitude.
Conflict of interest statement
Figures




Similar articles
-
Control of the firing patterns of vibrissa motoneurons by modulatory and phasic synaptic inputs: a modeling study.J Neurophysiol. 2010 May;103(5):2684-99. doi: 10.1152/jn.01016.2009. Epub 2010 Mar 3. J Neurophysiol. 2010. PMID: 20200122
-
Mixed mode oscillations in mouse spinal motoneurons arise from a low excitability state.J Neurosci. 2011 Apr 13;31(15):5829-40. doi: 10.1523/JNEUROSCI.6363-10.2011. J Neurosci. 2011. PMID: 21490224 Free PMC article.
-
Inhibition, Not Excitation, Drives Rhythmic Whisking.Neuron. 2016 Apr 20;90(2):374-87. doi: 10.1016/j.neuron.2016.03.007. Epub 2016 Mar 31. Neuron. 2016. PMID: 27041498 Free PMC article.
-
Anatomical loops and their electrical dynamics in relation to whisking by rat.Somatosens Mot Res. 1999;16(2):69-88. doi: 10.1080/08990229970528. Somatosens Mot Res. 1999. PMID: 10449057 Review.
-
Modulation of the intrinsic properties of motoneurons by serotonin.Curr Pharm Des. 2013;19(24):4371-84. doi: 10.2174/13816128113199990341. Curr Pharm Des. 2013. PMID: 23360270 Review.
Cited by
-
Mixed-mode oscillations in pyramidal neurons under antiepileptic drug conditions.PLoS One. 2017 Jun 7;12(6):e0178244. doi: 10.1371/journal.pone.0178244. eCollection 2017. PLoS One. 2017. PMID: 28591171 Free PMC article.
-
Understanding perspectives for mixed mode oscillations of the fractional neural network approaches to the analysis of neurophysiological data from the perspective of the observability of complex networks.Heliyon. 2024 Nov 26;10(23):e40659. doi: 10.1016/j.heliyon.2024.e40659. eCollection 2024 Dec 15. Heliyon. 2024. PMID: 39669132 Free PMC article.
-
Mixed-mode oscillations and population bursting in the pre-Bötzinger complex.Elife. 2016 Mar 14;5:e13403. doi: 10.7554/eLife.13403. Elife. 2016. PMID: 26974345 Free PMC article.
-
Diverse electrical responses in a network of fractional-order conductance-based excitable Morris-Lecar systems.Sci Rep. 2023 May 22;13(1):8215. doi: 10.1038/s41598-023-34807-3. Sci Rep. 2023. PMID: 37217514 Free PMC article.
-
Low-dimensional models of single neurons: a review.Biol Cybern. 2023 Jun;117(3):163-183. doi: 10.1007/s00422-023-00960-1. Epub 2023 Apr 15. Biol Cybern. 2023. PMID: 37060453 Review.
References
-
- Herfst LJ, Brecht M (2008) Whisker movements evoked by stimulation of single motor neurons in the facial nucleus of the rat. J Neurophysiol 99: 2821–2832. - PubMed
-
- Haidarliu S, Simony E, Golomb D, Ahissar E (2010) Muscle architecture in the mystacial pad of the rat. Anat Rec (Hoboken) 293: 1192–1206. - PubMed
-
- Berg RW, Kleinfeld D (2003) Rhythmic whisking by rat: retraction as well as protraction of the vibrissae is under active muscular control. J Neurophysiol 89: 104–117. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

