Blood vascular abnormalities in Rasa1(R780Q) knockin mice: implications for the pathogenesis of capillary malformation-arteriovenous malformation
- PMID: 25283357
- PMCID: PMC4258499
- DOI: 10.1016/j.ajpath.2014.08.018
Blood vascular abnormalities in Rasa1(R780Q) knockin mice: implications for the pathogenesis of capillary malformation-arteriovenous malformation
Abstract
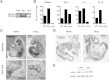
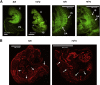
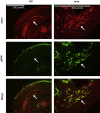
Capillary malformation-arteriovenous malformation (CM-AVM) is an autosomal dominant blood vascular (BV) disorder characterized by CM and fast flow BV lesions. Inactivating mutations of the RASA1 gene are the cause of CM-AVM in most cases. RASA1 is a GTPase-activating protein that acts as a negative regulator of the Ras small GTP-binding protein. In addition, RASA1 performs Ras-independent functions in intracellular signal transduction. Whether CM-AVM results from loss of an ability of RASA1 to regulate Ras or loss of a Ras-independent function of RASA1 is unknown. To address this, we generated Rasa1 knockin mice with an R780Q point mutation that abrogates RASA1 catalytic activity specifically. Homozygous Rasa1(R780Q/R780Q) mice showed the same severe BV abnormalities as Rasa1-null mice and died midgestation. This finding indicates that BV abnormalities in CM-AVM develop as a result of loss of an ability of RASA1 to control Ras activation and not loss of a Ras-independent function of this molecule. More important, findings indicate that inhibition of Ras signaling is likely to represent an effective means of therapy for this disease.
Copyright © 2014 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
RASA1 mutations and associated phenotypes in 68 families with capillary malformation-arteriovenous malformation.Hum Mutat. 2013 Dec;34(12):1632-41. doi: 10.1002/humu.22431. Epub 2013 Oct 10. Hum Mutat. 2013. PMID: 24038909
-
Somatic second hit mutation of RASA1 in vascular endothelial cells in capillary malformation-arteriovenous malformation.Eur J Med Genet. 2018 Jan;61(1):11-16. doi: 10.1016/j.ejmg.2017.10.004. Epub 2017 Oct 9. Eur J Med Genet. 2018. PMID: 29024832 Free PMC article.
-
RASA1 somatic mutation and variable expressivity in capillary malformation/arteriovenous malformation (CM/AVM) syndrome.Am J Med Genet A. 2016 Jun;170(6):1450-4. doi: 10.1002/ajmg.a.37613. Epub 2016 Mar 11. Am J Med Genet A. 2016. PMID: 26969842
-
RhoA activation-mediated vascular permeability in capillary malformation-arteriovenous malformation syndrome: a hypothesis.Drug Discov Today. 2021 Aug;26(8):1790-1793. doi: 10.1016/j.drudis.2020.12.012. Epub 2020 Dec 24. Drug Discov Today. 2021. PMID: 33358701 Review.
-
[Neonatal capillary malformation-arteriovenous malformation complicated with acute heart failure: a case report and literature review].Zhonghua Er Ke Za Zhi. 2020 Jul 2;58(7):591-595. doi: 10.3760/cma.j.cn112140-20200312-00221. Zhonghua Er Ke Za Zhi. 2020. PMID: 32605345 Review. Chinese.
Cited by
-
Brain arteriovenous malformation in hereditary hemorrhagic telangiectasia: Recent advances in cellular and molecular mechanisms.Front Hum Neurosci. 2022 Nov 24;16:1006115. doi: 10.3389/fnhum.2022.1006115. eCollection 2022. Front Hum Neurosci. 2022. PMID: 36504622 Free PMC article. Review.
-
EphrinB2-EphB4-RASA1 Signaling in Human Cerebrovascular Development and Disease.Trends Mol Med. 2019 Apr;25(4):265-286. doi: 10.1016/j.molmed.2019.01.009. Epub 2019 Feb 25. Trends Mol Med. 2019. PMID: 30819650 Free PMC article. Review.
-
Somatic Gain of KRAS Function in the Endothelium Is Sufficient to Cause Vascular Malformations That Require MEK but Not PI3K Signaling.Circ Res. 2020 Aug 28;127(6):727-743. doi: 10.1161/CIRCRESAHA.119.316500. Epub 2020 Jun 17. Circ Res. 2020. PMID: 32552404 Free PMC article.
-
Arteriovenous Cerebral High Flow Shunts in Children: From Genotype to Phenotype.Front Pediatr. 2022 Apr 25;10:871565. doi: 10.3389/fped.2022.871565. eCollection 2022. Front Pediatr. 2022. PMID: 35547535 Free PMC article.
-
RASA1-driven cellular export of collagen IV is required for the development of lymphovenous and venous valves in mice.Development. 2020 Dec 7;147(23):dev192351. doi: 10.1242/dev.192351. Development. 2020. PMID: 33144395 Free PMC article.
References
-
- Revencu N., Boon L.M., Mulliken J.B., Enjolras O., Cordisco M.R., Burrows P.E., Clapuyt P., Hammer F., Dubois J., Baselga E., Brancati F., Carder R., Quintal J.M., Dallapiccola B., Fischer G., Frieden I.J., Garzon M., Harper J., Johnson-Patel J., Labreze C., Martorell L., Paltiel H.J., Pohl A., Prendiville J., Quere I., Siegel D.H., Valente E.M., Van Hagen A., Van Hest L., Vaux K.K., Vicente A., Weibel L., Chitayat D., Vikkula M. Parkes Weber syndrome, vein of Galen aneurysmal malformation, and other fast-flow vascular anomalies are caused by RASA1 mutations. Hum Mutat. 2008;29:959–965. - PubMed
-
- Revencu N., Boon L.M., Mendola A., Cordisco M.R., Dubois J., Clapuyt P. RASA1 mutations and associated phenotypes in 68 families with capillary malformation-arteriovenous malformation. Hum Mutat. 2013;34:1632–1641. - PubMed
-
- Bayrak-Toydemir P, Stevenson D: RASA1-Related Disorders. Edited by RA Pagon, MP Adam, HH Ardinger, et al. In GeneReviews [Internet]. Copyright University of Washington, Seattle. 2011. Available at http://www.ncbi.nlm.nih.gov/books/NBK52764, last revised December 19, 2013
-
- de Wijn R.S., Oduber C.E., Breugem C.C., Alders M., Hennekam R.C., van der Horst C.M. Phenotypic variability in a family with capillary malformations caused by a mutation in the RASA1 gene. Eur J Med Genet. 2012;55:191–195. - PubMed
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

