Experimental scleral cross-linking increases glaucoma damage in a mouse model
- PMID: 25285424
- PMCID: PMC4254118
- DOI: 10.1016/j.exer.2014.08.016
Experimental scleral cross-linking increases glaucoma damage in a mouse model
Abstract
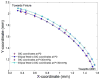
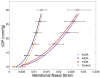
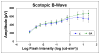
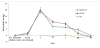
The purpose of this study was to assess the effect of a scleral cross-linking agent on susceptibility to glaucoma damage in a mouse model.CD1 mice underwent 3 subconjunctival injections of 0.5 M glyceraldehyde (GA) in 1 week, then had elevated intraocular pressure (IOP) induced by bead injection. Degree of cross-linking was measured by enzyme-linked immunosorbent assay (ELISA), scleral permeability was measured by fluorescence recovery after photobleaching (FRAP), and the mechanical effects of GA exposure were measured by inflation testing. Control mice had buffer injection or no injection in 2 separate glaucoma experiments. IOP was monitored by Tonolab and retinal ganglion cell (RGC) loss was measured by histological axon counting. To rule out undesirable effects of GA, we performed electroretinography and detailed histology of the retina. GA exposure had no detectable effects on RGC number, retinal structure or function either histologically or electrophysiologically. GA increased cross-linking of sclera by 37% in an ELISA assay, decreased scleral permeability (FRAP, p = 0.001), and produced a steeper pressure-strain behavior by in vitro inflation testing. In two experimental glaucoma experiments, GA-treated eyes had greater RGC axon loss from elevated IOP than either buffer-injected or control eyes, controlling for level of IOP exposure over time (p = 0.01, and 0.049, multivariable regression analyses). This is the first report that experimental alteration of the sclera, by cross-linking, increases susceptibility to RGC damage in mice.
Keywords: Collagen; Cross-linking; Extracellular matrix; Glaucoma; Glyceraldehyde; Mouse; Retinal ganglion cell; Sclera.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
Studies of scleral biomechanical behavior related to susceptibility for retinal ganglion cell loss in experimental mouse glaucoma.Invest Ophthalmol Vis Sci. 2013 Mar 11;54(3):1767-80. doi: 10.1167/iovs.12-10952. Invest Ophthalmol Vis Sci. 2013. PMID: 23404116 Free PMC article.
-
Mice with an induced mutation in collagen 8A2 develop larger eyes and are resistant to retinal ganglion cell damage in an experimental glaucoma model.Mol Vis. 2012;18:1093-106. Epub 2012 May 1. Mol Vis. 2012. PMID: 22701298 Free PMC article.
-
Losartan Treatment Protects Retinal Ganglion Cells and Alters Scleral Remodeling in Experimental Glaucoma.PLoS One. 2015 Oct 27;10(10):e0141137. doi: 10.1371/journal.pone.0141137. eCollection 2015. PLoS One. 2015. PMID: 26505191 Free PMC article.
-
Development of diagnostic and treatment strategies for glaucoma through understanding and modification of scleral and lamina cribrosa connective tissue.Cell Tissue Res. 2013 Aug;353(2):231-44. doi: 10.1007/s00441-013-1603-0. Epub 2013 Mar 28. Cell Tissue Res. 2013. PMID: 23535950 Free PMC article. Review.
-
The contribution of the sclera and lamina cribrosa to the pathogenesis of glaucoma: Diagnostic and treatment implications.Prog Brain Res. 2015;220:59-86. doi: 10.1016/bs.pbr.2015.04.003. Epub 2015 Jul 2. Prog Brain Res. 2015. PMID: 26497785 Review.
Cited by
-
Management of experimental trabeculectomy filtering blebs via crosslinking of the scleral flap inhibited vascularization.Graefes Arch Clin Exp Ophthalmol. 2024 May;262(5):1507-1517. doi: 10.1007/s00417-023-06306-8. Epub 2023 Nov 9. Graefes Arch Clin Exp Ophthalmol. 2024. PMID: 37943331
-
In Vivo Small Molecule Delivery to the Optic Nerve in a Rodent Model.Sci Rep. 2018 Mar 13;8(1):4453. doi: 10.1038/s41598-018-22737-4. Sci Rep. 2018. PMID: 29535357 Free PMC article.
-
High-Resolution, Three-Dimensional Reconstruction of the Outflow Tract Demonstrates Segmental Differences in Cleared Eyes.Invest Ophthalmol Vis Sci. 2018 May 1;59(6):2371-2380. doi: 10.1167/iovs.17-23075. Invest Ophthalmol Vis Sci. 2018. PMID: 29847643 Free PMC article.
-
Collagen Structural Changes in Rat Tarsus After Crosslinking.Transl Vis Sci Technol. 2021 Apr 29;10(5):3. doi: 10.1167/tvst.10.5.3. Transl Vis Sci Technol. 2021. PMID: 34003976 Free PMC article.
-
Age-Related Changes in Quantitative Strain of Mouse Astrocytic Lamina Cribrosa and Peripapillary Sclera Using Confocal Microscopy in an Explant Model.Invest Ophthalmol Vis Sci. 2018 Oct 1;59(12):5157-5166. doi: 10.1167/iovs.18-25111. Invest Ophthalmol Vis Sci. 2018. PMID: 30372742 Free PMC article.
References
-
- Avetisov ES, Savitskaya NF, Vinetskaya MI, Iomdina EN. A study of biochemical and biomechanical qualities of normal and myopic eye sclera in humans of different age groups. Metab Pediatr Syst Ophthalmol. 1984;7:183–188. - PubMed
-
- Backhouse S, Phillips JR. Effect of induced myopia on scleral myofibroblasts and in vivo ocular biomechanical compliance in the guinea pig. Invest Ophthalmol Vis Sci. 2010;51:6162–6171. - PubMed
-
- Bengtsson B, Heijl A. Diurnal IOP fluctuation: not an independent risk factor for glaucomatous visual field loss in high-risk ocular hypertension. Graefe’s Arch Clin Exper Ophthalmol. 2005;243:513–8. - PubMed
-
- Boland MV, Quigley HA. Risk factors and open-angle glaucoma: concepts and applications. J Glaucoma. 2007;16:406–418. - PubMed
-
- Burgoyne CF, Downs JC, Bellezza AJ, Suh J-KF, Hart RT. The optic nerve head as a biomechanical structure: a new paradigm for understanding the role of IOP-related stress and strain in the pathophysiology of glaucomatous optic nerve head damage. Prog Ret Eye Res. 2005;24:39–73. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

