Enhancing ubiquitin crystallization through surface-entropy reduction
- PMID: 25286958
- PMCID: PMC4188098
- DOI: 10.1107/S2053230X14019244
Enhancing ubiquitin crystallization through surface-entropy reduction
Abstract
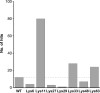
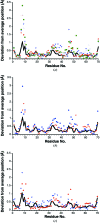
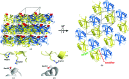
Ubiquitin has many attributes suitable for a crystallization chaperone, including high stability and ease of expression. However, ubiquitin contains a high surface density of lysine residues and the doctrine of surface-entropy reduction suggests that these lysines will resist participating in packing interactions and thereby impede crystallization. To assess the contributions of these residues to crystallization behavior, each of the seven lysines of ubiquitin was mutated to serine and the corresponding single-site mutant proteins were expressed and purified. The behavior of these seven mutants was then compared with that of the wild-type protein in a 384-condition crystallization screen. The likelihood of obtaining crystals varied by two orders of magnitude within this set of eight proteins. Some mutants crystallized much more readily than the wild type, while others crystallized less readily. X-ray crystal structures were determined for three readily crystallized variants: K11S, K33S and the K11S/K63S double mutant. These structures revealed that the mutant serine residues can directly promote crystallization by participating in favorable packing interactions; the mutations can also exert permissive effects, wherein crystallization appears to be driven by removal of the lysine rather than by addition of a serine. Presumably, such permissive effects reflect the elimination of steric and electrostatic barriers to crystallization.
Keywords: surface-entropy reduction; ubiquitin.
Figures
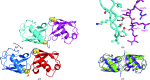
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

