A Putative Transcription Factor pcs1 Positively Regulates Both Conidiation and Sexual Reproduction in the Cereal Pathogen Fusarium graminearum
- PMID: 25289009
- PMCID: PMC4181113
- DOI: 10.5423/PPJ.OA.04.2014.0037
A Putative Transcription Factor pcs1 Positively Regulates Both Conidiation and Sexual Reproduction in the Cereal Pathogen Fusarium graminearum
Abstract
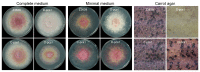
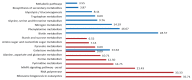
The plant pathogen Fusarium graminearum causes Fusarium head blight in cereal crops and produces mycotoxins that are harmful to animals and humans. For the initiation and spread of disease, asexual and sexual reproduction is required. Therefore, studies on fungal reproduction contribute to the development of new methods to control and maintain the fungal population. Screening a previously generated transcription factor mutant collection, we identified one putative C2H2 zinc-finger transcription factor, pcs1, which is required for both sexual and asexual reproduction. Deleting pcs1 in F. graminearum resulted in a dramatic reduction in conidial production and a complete loss of sexual reproduction. The pathways and gene ontology of pcs1-dependent genes from microarray experiments showed that several G-protein related pathways, oxidase activity, ribosome biogenesis, and RNA binding and processing were highly enriched, suggesting that pcs1 is involved in several different biological processes. Further, overexpression of pcs1 increased conidial production and resulted in earlier maturation of ascospores compared to the wild-type strain. Additionally, the vegetative growth of the overexpression mutants was decreased in nutrient-rich conditions but was not different from the wild-type strain in nutrient-poor conditions. Overall, we discovered that the pcs1 transcription factor positively regulates both conidiation and sexual reproduction and confers nutrient condition-dependent vegetative growth.
Keywords: Conidiation; Fusarium graminearum; Gibberella zeae; sexual reproduction; transcription factor.
Figures



Similar articles
-
Recent advances in genes involved in secondary metabolite synthesis, hyphal development, energy metabolism and pathogenicity in Fusarium graminearum (teleomorph Gibberella zeae).Biotechnol Adv. 2014 Mar-Apr;32(2):390-402. doi: 10.1016/j.biotechadv.2013.12.007. Epub 2014 Jan 2. Biotechnol Adv. 2014. PMID: 24389085 Review.
-
FgEaf6 regulates virulence, asexual/sexual development and conidial septation in Fusarium graminearum.Curr Genet. 2020 Jun;66(3):517-529. doi: 10.1007/s00294-019-01043-0. Epub 2019 Nov 14. Curr Genet. 2020. PMID: 31728616
-
AbaA regulates conidiogenesis in the ascomycete fungus Fusarium graminearum.PLoS One. 2013 Sep 10;8(9):e72915. doi: 10.1371/journal.pone.0072915. eCollection 2013. PLoS One. 2013. PMID: 24039821 Free PMC article.
-
FgFlbD regulates hyphal differentiation required for sexual and asexual reproduction in the ascomycete fungus Fusarium graminearum.J Microbiol. 2014 Nov;52(11):930-9. doi: 10.1007/s12275-014-4384-6. Epub 2014 Oct 3. J Microbiol. 2014. PMID: 25277408
-
On the trail of a cereal killer: recent advances in Fusarium graminearum pathogenomics and host resistance.Mol Plant Pathol. 2012 May;13(4):399-413. doi: 10.1111/j.1364-3703.2011.00762.x. Epub 2011 Nov 20. Mol Plant Pathol. 2012. PMID: 22098555 Free PMC article. Review.
Cited by
-
Cooperative interactions between seed-borne bacterial and air-borne fungal pathogens on rice.Nat Commun. 2018 Jan 2;9(1):31. doi: 10.1038/s41467-017-02430-2. Nat Commun. 2018. PMID: 29295978 Free PMC article.
-
FgSsn3 kinase, a component of the mediator complex, is important for sexual reproduction and pathogenesis in Fusarium graminearum.Sci Rep. 2016 Mar 2;6:22333. doi: 10.1038/srep22333. Sci Rep. 2016. PMID: 26931632 Free PMC article.
-
The Role of Chromatin and Transcriptional Control in the Formation of Sexual Fruiting Bodies in Fungi.Microbiol Mol Biol Rev. 2022 Dec 21;86(4):e0010422. doi: 10.1128/mmbr.00104-22. Epub 2022 Nov 21. Microbiol Mol Biol Rev. 2022. PMID: 36409109 Free PMC article. Review.
References
-
- Bowden RL, Leslie JF. Sexual recombination in Gibberella zeae. Phytopathology. 1999;89:182–188. - PubMed
-
- Capellini R, Peterson J. Macroconidium formation in submerged cultures by non-sporulating strains of Gibberella zeae. Mycologia. 1965;57:962–966.
-
- Fernando WGD, Paulitz TC, Seaman WL, Dutilleul P, Miller JD. Head blight gradients caused by Gibberella zeae from area sources of inoculums in wheat field plots. Phytopathology. 1997;87:414–421. - PubMed
-
- Guenther JC, Trail F. The development and differentiation of Gibberella zeae (anamorph: Fusarium graminearum) during colonization of wheat. Mycologia. 2005;97:229–237. - PubMed
-
- Hallen HE, Huebner M, Shiu S-H, Güldener U, Trail F. Gene expression shifts during perithecium development in Gibberella zeae (anamorph Fusarium graminearum), with particular emphasis on ion transport proteins. Fungal Genet Biol. 2007;44:1146–1156. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

