First-in-man study of western reserve strain oncolytic vaccinia virus: safety, systemic spread, and antitumor activity
- PMID: 25292189
- PMCID: PMC4426804
- DOI: 10.1038/mt.2014.194
First-in-man study of western reserve strain oncolytic vaccinia virus: safety, systemic spread, and antitumor activity
Abstract
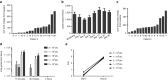
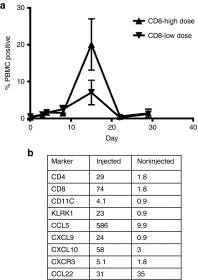
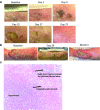
Oncolytic viral therapy utilizes a tumor-selective replicating virus which preferentially infects and destroys cancer cells and triggers antitumor immunity. The Western Reserve strain of vaccinia virus (VV) is the most virulent strain of VV in animal models and has been engineered for tumor selectivity through two targeted gene deletions (vvDD). We performed the first-in-human phase 1, intratumoral dose escalation clinical trial of vvDD in 16 patients with advanced solid tumors. In addition to safety, we evaluated signs of vvDD replication and spread to distant tumors, pharmacokinetics and pharmacodynamics, clinical and immune responses to vvDD. Dose escalation proceeded without dose-limiting toxicities to a maximum feasible dose of 3 × 10(9) pfu. vvDD replication in tumors was reproducible. vvDD genomes and/or infectious particles were recovered from injected (n = 5 patients) and noninjected (n = 2 patients) tumors. At the two highest doses, vvDD genomes were detected acutely in blood in all patients while delayed re-emergence of vvDD genomes in blood was detected in two patients. Fifteen of 16 patients exhibited late symptoms, consistent with ongoing vvDD replication. In summary, intratumoral injection of the oncolytic vaccinia vvDD was well-tolerated in patients and resulted in selective infection of injected and noninjected tumors and antitumor activity.
Figures

Similar articles
-
Antiangiogenic arming of an oncolytic vaccinia virus enhances antitumor efficacy in renal cell cancer models.J Virol. 2010 Jan;84(2):856-66. doi: 10.1128/JVI.00692-09. Epub 2009 Nov 11. J Virol. 2010. PMID: 19906926 Free PMC article.
-
Phase 1 Study of Intravenous Oncolytic Poxvirus (vvDD) in Patients With Advanced Solid Cancers.Mol Ther. 2016 Aug;24(8):1492-501. doi: 10.1038/mt.2016.101. Epub 2016 May 16. Mol Ther. 2016. PMID: 27203445 Free PMC article. Clinical Trial.
-
Elucidating mechanisms of antitumor immunity mediated by live oncolytic vaccinia and heat-inactivated vaccinia.J Immunother Cancer. 2021 Sep;9(9):e002569. doi: 10.1136/jitc-2021-002569. J Immunother Cancer. 2021. PMID: 34593618 Free PMC article.
-
Vaccinia virus, a promising new therapeutic agent for pancreatic cancer.Immunotherapy. 2015;7(12):1249-58. doi: 10.2217/imt.15.90. Epub 2015 Nov 23. Immunotherapy. 2015. PMID: 26595180 Free PMC article. Review.
-
Herpes simplex virus oncolytic vaccine therapy in melanoma.Expert Opin Biol Ther. 2010 Jul;10(7):1145-53. doi: 10.1517/14712598.2010.495383. Expert Opin Biol Ther. 2010. PMID: 20515292 Review.
Cited by
-
Optimizing Oncolytic Viral Design to Enhance Antitumor Efficacy: Progress and Challenges.Cancers (Basel). 2020 Jun 26;12(6):1699. doi: 10.3390/cancers12061699. Cancers (Basel). 2020. PMID: 32604787 Free PMC article. Review.
-
Oncolytic properties of non-vaccinia poxviruses.Oncotarget. 2018 Nov 13;9(89):35891-35906. doi: 10.18632/oncotarget.26288. eCollection 2018 Nov 13. Oncotarget. 2018. PMID: 30542506 Free PMC article.
-
lncRNA UCA1-Mediated Cdc42 Signaling Promotes Oncolytic Vaccinia Virus Cell-to-Cell Spread in Ovarian Cancer.Mol Ther Oncolytics. 2019 Mar 26;13:35-48. doi: 10.1016/j.omto.2019.03.003. eCollection 2019 Jun 28. Mol Ther Oncolytics. 2019. PMID: 31011626 Free PMC article.
-
Design and Engineering of Deimmunized Vaccinia Viral Vectors.Biomedicines. 2020 Nov 11;8(11):491. doi: 10.3390/biomedicines8110491. Biomedicines. 2020. PMID: 33187060 Free PMC article. Review.
-
Cytokine-armed vaccinia virus promotes cytotoxicity toward pancreatic carcinoma cells via activation of human intermediary CD56dimCD16dim natural killer cells.Int J Cancer. 2025 Feb 1;156(3):638-651. doi: 10.1002/ijc.35209. Epub 2024 Oct 14. Int J Cancer. 2025. PMID: 39400317 Free PMC article.
References
-
- Martuza RL, Malick A, Markert JM, Ruffner KL, Coen DM. Experimental therapy of human glioma by means of a genetically engineered virus mutant. Science. 1991;252:854–856. - PubMed
-
- Kirn D. Clinical research results with dl1520 (Onyx-015), a replication-selective adenovirus for the treatment of cancer: what have we learned. Gene Ther. 2001;8:89–98. - PubMed
-
- Nemunaitis J, Cunningham C, Buchanan A, Blackburn A, Edelman G, Maples P, et al. Intravenous infusion of a replication-selective adenovirus (ONYX-015) in cancer patients: safety, feasibility and biological activity. Gene Ther. 2001;8:746–759. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

