Nicotinic and muscarinic acetylcholine receptors shape ganglion cell response properties
- PMID: 25298382
- PMCID: PMC4294565
- DOI: 10.1152/jn.00405.2014
Nicotinic and muscarinic acetylcholine receptors shape ganglion cell response properties
Abstract
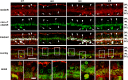
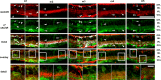
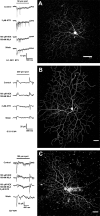
The purpose of this study was to evaluate the expression patterns of nicotinic and muscarinic ACh receptors (nAChRs and mAChRs, respectively) in relation to one another and to understand their effects on rabbit retinal ganglion cell response properties. Double-label immunohistochemistry revealed labeled inner-retinal cell bodies and complex patterns of nAChR and mAChR expression in the inner plexiform layer. Specifically, the expression patterns of m1, m4, and m5 muscarinic receptors overlapped with those of non-α7 and α7 nicotinic receptors in presumptive amacrine and ganglion cells. There was no apparent overlap in the expression patterns of m2 muscarinic receptors with α7 nicotinic receptors or of m3 with non-α7 nicotinic receptors. Patch-clamp recordings demonstrated cell type-specific effects of nicotinic and muscarinic receptor blockade. Muscarinic receptor blockade enhanced the center responses of brisk-sustained/G4 On and G4 Off ganglion cells, whereas nicotinic receptor blockade suppressed the center responses of G4 On-cells near the visual streak but enhanced the center responses of nonstreak G4 On-cells. Blockade of muscarinic or nicotinic receptors suppressed the center responses of brisk-sustained Off-cells and the center light responses of subsets of brisk-transient/G11 On- and Off-cells. Only nicotinic blockade affected the center responses of G10 On-cells and G5 Off-cells. These data indicate that physiologically and morphologically identified ganglion cell types have specific patterns of AChR expression. The cholinergic receptor signatures of these cells may have implications for understanding visual defects in disease states that result from decreased ACh availability.
Keywords: cholinergic agonists; cholinergic antagonists; muscarinic acetylcholine receptors; nicotinic acetylcholine receptors; retinal circuitry.
Copyright © 2015 the American Physiological Society.
Figures






Similar articles
-
Strychnine, but not PMBA, inhibits neuronal nicotinic acetylcholine receptors expressed by rabbit retinal ganglion cells.Vis Neurosci. 2007 Jul-Aug;24(4):503-11. doi: 10.1017/S0952523807070241. Vis Neurosci. 2007. PMID: 17900376
-
Properties of cholinergic responses in neurons in the intermediate grey layer of rat superior colliculus.Eur J Neurosci. 2006 Dec;24(11):3096-108. doi: 10.1111/j.1460-9568.2006.05190.x. Eur J Neurosci. 2006. PMID: 17156371
-
Acetylcholine receptors in the retinas of the α7 nicotinic acetylcholine receptor knockout mouse.Mol Vis. 2014 Sep 20;20:1328-56. eCollection 2014. Mol Vis. 2014. PMID: 25352741 Free PMC article.
-
Muscarinic and nicotinic acetylcholine receptor agonists: current scenario in Alzheimer's disease therapy.J Pharm Pharmacol. 2018 Aug;70(8):985-993. doi: 10.1111/jphp.12919. Epub 2018 Apr 16. J Pharm Pharmacol. 2018. PMID: 29663387 Review.
-
Regulation of Immune Functions by Non-Neuronal Acetylcholine (ACh) via Muscarinic and Nicotinic ACh Receptors.Int J Mol Sci. 2021 Jun 24;22(13):6818. doi: 10.3390/ijms22136818. Int J Mol Sci. 2021. PMID: 34202925 Free PMC article. Review.
Cited by
-
Muscarinic Acetylcholine Receptors in the Retina-Therapeutic Implications.Int J Mol Sci. 2021 May 8;22(9):4989. doi: 10.3390/ijms22094989. Int J Mol Sci. 2021. PMID: 34066677 Free PMC article. Review.
-
Starburst amacrine cells amplify optogenetic visual restoration through gap junctions.Mol Ther Methods Clin Dev. 2023 May 12;30:1-13. doi: 10.1016/j.omtm.2023.05.011. eCollection 2023 Sep 14. Mol Ther Methods Clin Dev. 2023. PMID: 37324975 Free PMC article.
-
Retinal changes in the Tg-SwDI mouse model of Alzheimer's disease.Neuroscience. 2017 Jun 23;354:43-53. doi: 10.1016/j.neuroscience.2017.04.021. Epub 2017 Apr 25. Neuroscience. 2017. PMID: 28450267 Free PMC article.
-
Bipolar Cell Type-Specific Expression and Conductance of Alpha-7 Nicotinic Acetylcholine Receptors in the Mouse Retina.Invest Ophthalmol Vis Sci. 2019 Apr 1;60(5):1353-1361. doi: 10.1167/iovs.18-25753. Invest Ophthalmol Vis Sci. 2019. PMID: 30934054 Free PMC article.
-
Unveiling the Neural Environment in Cancer: Exploring the Role of Neural Circuit Players and Potential Therapeutic Strategies.Cells. 2023 Aug 3;12(15):1996. doi: 10.3390/cells12151996. Cells. 2023. PMID: 37566075 Free PMC article. Review.
References
-
- Albuquerque EX, Alkondon M, Pereira EF, Castro NG, Schrattenholz A, Barbosa CT, Bonfante-Cabarcas R, Aracava Y, Eisenberg HM, Maelicke A. Properties of neuronal nicotinic acetylcholine receptors: pharmacological characterization and modulation of synaptic function. J Pharmacol Exp Ther 280: 1117–1136, 1997. - PubMed
-
- Alhomida AS, Al-Rajhi AA, Kamal MA, Al-Jafari AA. Kinetic analysis of the toxicological effect of tacrine (Cognex) on human retinal acetylcholinesterase activity. Toxicology 147: 33–39, 2000. - PubMed
-
- Alkondon M, Pereira EF, Cortes WS, Maelicke A, Albuquerque EX. Choline is a selective agonist of alpha7 nicotinic acetylcholine receptors in the rat brain neurons. Eur J Neurosci 9: 2734–2742, 1997. - PubMed
-
- Alkondon M, Pereira EF, Wonnacott S, Albuquerque EX. Blockade of nicotinic currents in hippocampal neurons defines methyllycaconitine as a potent and specific receptor antagonist. Mol Pharmacol 41: 802–808, 1992. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

